BLOG > mRNA & LNP > White paper: Ionizable lipids for mRNA-LNP and siRNA-LNP
This white paper examines the development of Lipid Nanoparticles (LNP) formulations for mRNA delivery, focusing on innovations at OZ Biosciences. We present data demonstrating how ionizable lipid composition influences transfection efficiency across various cell lines and affects in vivo biodistribution, with particular emphasis on intra venous administration in murine models.
Introduction
The emergence of mRNA-based therapeutics has created an urgent need for efficient delivery systems capable of protecting nucleic acids from degradation while ensuring cellular uptake and endosomal escape. Lipid Nanoparticles have proven to be the most successful delivery platform, as evidenced by their use in COVID-19 vaccines. However, achieving cell-type specific delivery and optimal biodistribution remains challenging.
OZ Biosciences has developed a series of LNP formulations that demonstrate differential transfection efficiency across cell types in culture and tissues in living organisms. This white paper presents comparative data on how various ionizable lipid compositions affect mRNA delivery both in vitro and in vivo settings, with special focus on intravenous injection as a route of injection for targeting various organs.
LNP Composition and Design Principles
Standard LNP formulations consist of four key components in varying doses. The complexing lipid, in our case a proprietary ionizable lipid, counts for 40-50 mol%; the helper phospholipid that stabilizes and structures the LNP, for 10-20 mol%; the cholesterol that ensures the rigidity and integrity of the LNP, represents 35-45 mol% and finally, the PEG-lipid that increases the bio-circulation represents 1-3 mol%.
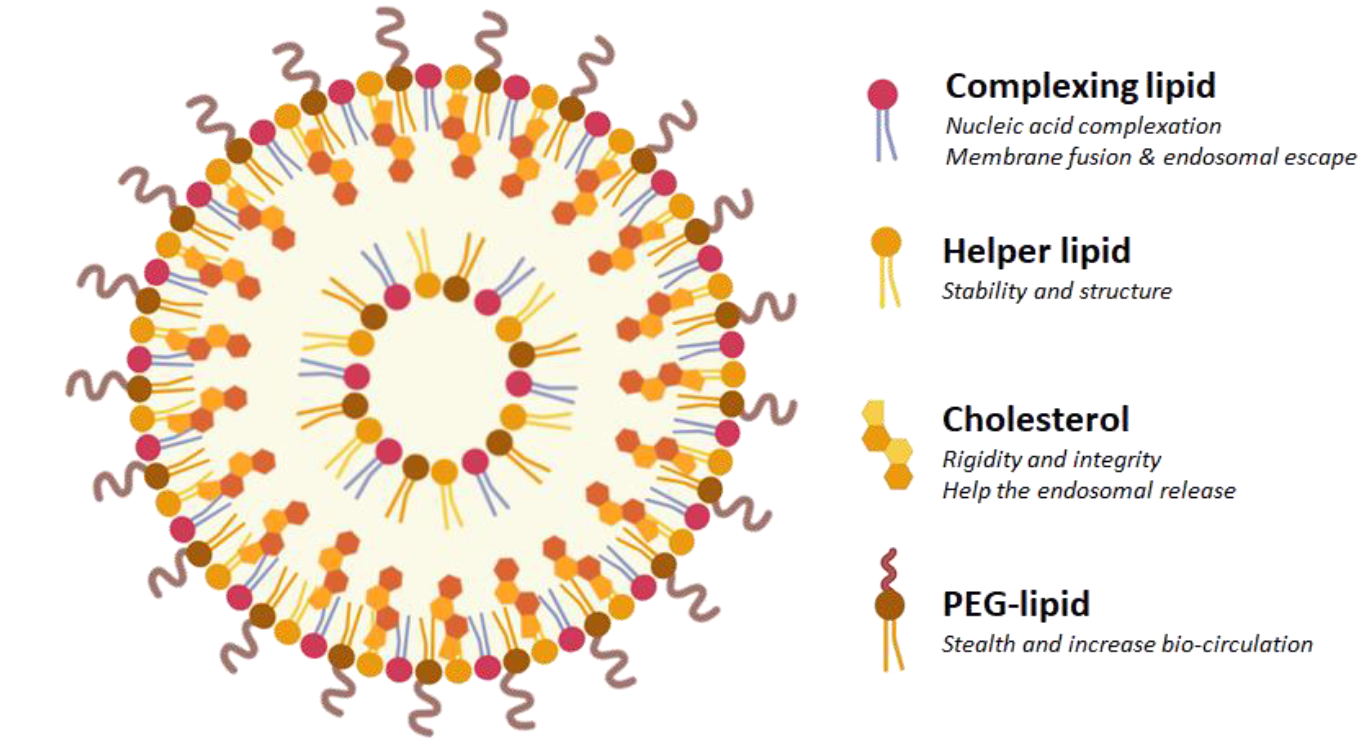
Figure 1: Schematic representation of a lipid nanoparticle composed by a mixture of four chemical components: complexing (ionizable) lipid, helper phospholipid, cholesterol and stealth-lipid. Each compound holds its own specific function and is present at a defined concentration.
The ionizable lipid component is critical for endosomal escape and help to determine much of the LNP's cell-type specificity and biodistribution profile.
The formulation is done by microfluidic or jet mixer where the active ingredient is mixed with lipids before adjustment by dilution. The system of mixing depends on the application and the volume required.
All our lipids are designed based on our patented structure that allows to change the type and length of the carbon tail, the polar head and the bio-inspired molecular template. Each created LNP has thus a specific size and electrical charge, as well as a defined mRNA encapsulation rate that are validated through extensive quality controls (Table 1).
| Items | Specification | Standard QC | Superior Grade QC |
| Identity | Size | ✔ | ✔ |
| Charge | ✔ | ✔ | |
| Content | Encapsulation efficiency | ✔ | ✔ |
| RNA concentration | ✔ | ✔ | |
| Safety | Sterility | ✔ | ✔ |
| Endotoxin | ✔ | ||
| Mycoplasma detection | ✔ | ||
| Characterization | Lipid content | ✔ |
Table 1: Quality controls depend on the grade of the required LNP. While Standard QC includes the validation of size, charge, encapsulation efficiency, RNA concentration and sterility, Superior Grade QC extends its checks to additional aspects related to endotoxin and Mycoplasma detection as well as the dosage of lipids. Superior Grade QC thus offers a more comprehensive evaluation to ensure superior quality and safety.
Rationale
The role of ionizable lipids in cell-type specificity
The influence of ionizable lipids on transfection efficiency depends on multiple factors related to cell origin and characteristics. Primary cells and cell lines from different tissues exhibit varying membrane compositions, endocytic pathways, and metabolic activities. For instance, hepatocyte-derived cells seem to show preferential uptake of LNPs containing ionizable lipids with pKa values between 6.2-6.8. The territorial origin of cells also influences their response; cells from metabolically active tissues (liver, kidney) generally show higher transfection rates compared to cells from immune-privileged sites (brain, testes). This specificity may be attributed to differences in apolipoprotein binding patterns, which could be influenced by the ionizable lipid structure and surface charge distribution of the LNPs.
The role of ionizable Lipids in territorial targeting depending on the injection site
The nature of the ionizable lipids profoundly impacts LNP biodistribution in tissue targeting depending on the route of injection. Following intraperitoneal injection, LNPs first encounter peritoneal fluid and resident immune cells before systemic absorption. Intramuscular injections favor local muscle cell transfection with lipids containing unsaturated tails, whereas intravenous administration requires lipids that promote ApoE binding for hepatic targeting. The injection site essentially determines the first biological barriers encountered, and optimal ionizable lipid selection must account for these route-specific challenges to achieve desired tissue targeting.
Results
Ionizable lipid for in vitro mRNA delivery
We have demonstrated that depending on their composition or concentration in ionizable lipid or associated components, LNPs present different affinity to efficiently transfect cells from various origins. mRNA-GFP encoding for green fluorescent protein was encapsulated in LNPs formulated with ionizable lipids issued from our proprietary catalog. Various cell types from different origins were then transfected and GFP expression was monitored 24H later by fluorescence microscopy and Flow Cytometry.
The results confirmed that the ionizable lipids have a direct influence on the transfection, depending on the territory from which they originate (Table 2). LNP SS22 or LNP SS23 will be more recommended for dendritic cells, monocytes, macrophages and PBMC. Both of them are also good candidate for T cells as well as LNP SS12 whereas B cells will be more prone to be transfected by LNP SS18. Hepatocytes on the other hand would prefer SS18 or SS22, and SS23 or FP105 would be recommended for leukemia cells. Finally, transfection of common cell lines would be more efficient with LNP SS12 or SS18.
| Cell Type | Origin | Standard QC | Ionizable lipid recommended |
| B cells | human |
Raji U266 |
SS18 |
| Common cell lines |
human rodent |
HEK293 CHO-K1 |
SS12 | SS18 |
| Dendritic cells | mouse | DC2.4 | SS22 | SS23 |
| Hépatocytes | human | HuH7 | SS18 | SS22 |
| Leukaemia | human | MOLT4 | FP105 | SS23 |
| Lung cells | human | A549 | SS22 | FP105 |
| Macrophages | mouse |
RAW264.7 J774 |
SS22 | SS23 |
| Monocytes | human |
THP-1 U937 |
SS22 | SS23 |
| PBMC | human | PBMC | SS22 | SS23 |
| T cells |
human mouse |
HPB-ALL Jurkat TK-1 |
SS12 | SS 22 | SS23 |
Table 2: Recommended ionizable lipids to use as complexing lipid for LNP formulation depending on cell type and origin. Various types of cell lines from different origins were transfected with mRNA-LUC or -GFP encapsulated in LNP formulated with OZB catalog of ionizable lipids. The best candidate(s) for each cell type were chosen according to the efficiency of transfection.
We can then spot for each cell type which LNP works the best to draw a pattern allowing to choose more easily which lipid to recommend for which application (Table 3):
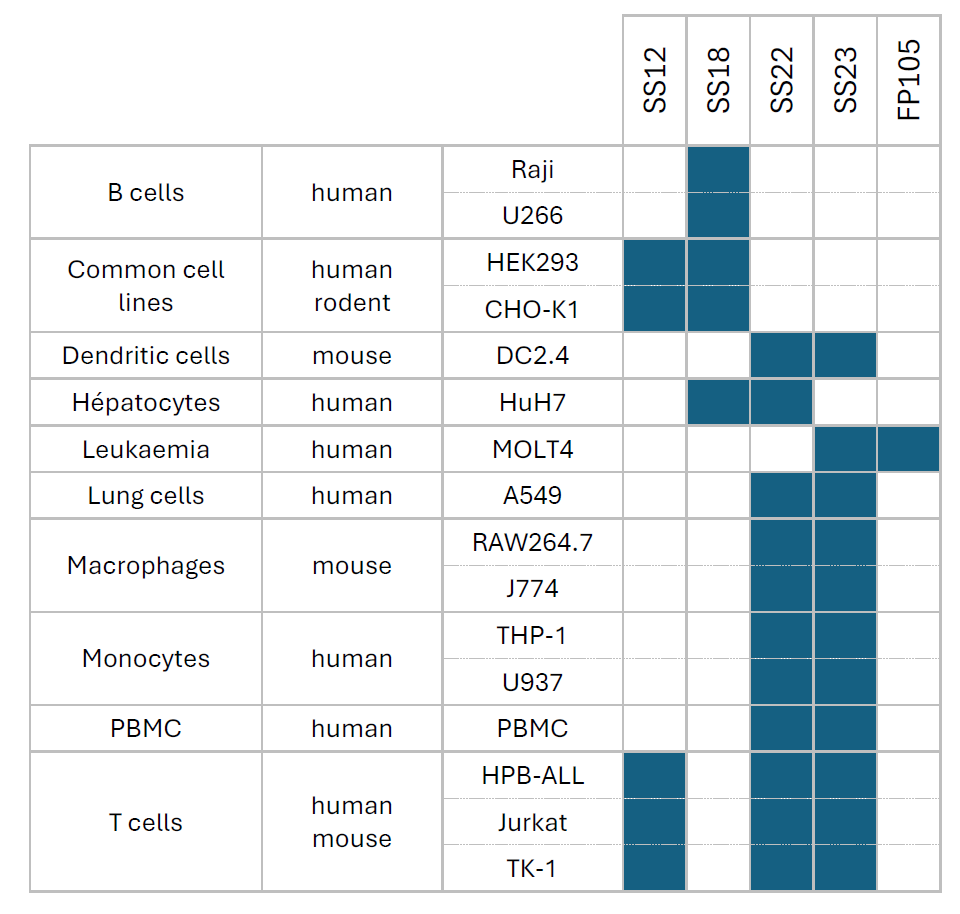
Table 3: Choice of ionizable lipid for mRNA-LNP formulation depending on the cell type and territory
Ionizable lipid for in vivo mRNA delivery
To evaluate the organ-targeting capabilities of our ionizable lipid library, mRNA encoding Firefly Luciferase (F-Luc mRNA #MRNA16-100) was encapsulated in lipid nanoparticles (LNPs) formulated with a panel of proprietary ionizable lipids. All formulations were administrated intravenously to mice, and luciferase expression was monitored 6 hours post-injection by bioluminescence imaging following intravenous injection of D-Luciferin. Both whole-body imaging (ventral and dorsal views) and ex vivo organ imaging were performed to accurately map the biodistribution of each formulation at the tissue level.
The experimental design revealed that the choice of ionizable lipid, as well as its concentration and the nature of the associated lipid components, is a primary determinant of LNP tropism following systemic delivery.
Depending on the formulation of the LNP, mRNA delivery could be directed preferentially to a single organ, distributed broadly across multiple tissues or finely tuned by adjusting the lipid composition.
a. LNP Multi-organ, a formulation to transfect the whole body
The LNP Multi-organ demonstrated a whole body distribution following intravenous administration. Luminescence signal was distributed across multiple anatomical regions (Figure 2A). Ex vivo organ imaging confirmed that luciferase expression was detectable across all analyzed organs reflecting the broad biodistribution. Quantification of the bioluminescence signal confirmed the broad distribution (Figure 2B).
This multi-organ formulation is particularly suited for applications requiring systemic mRNA delivery with simultaneous expression across several tissues.
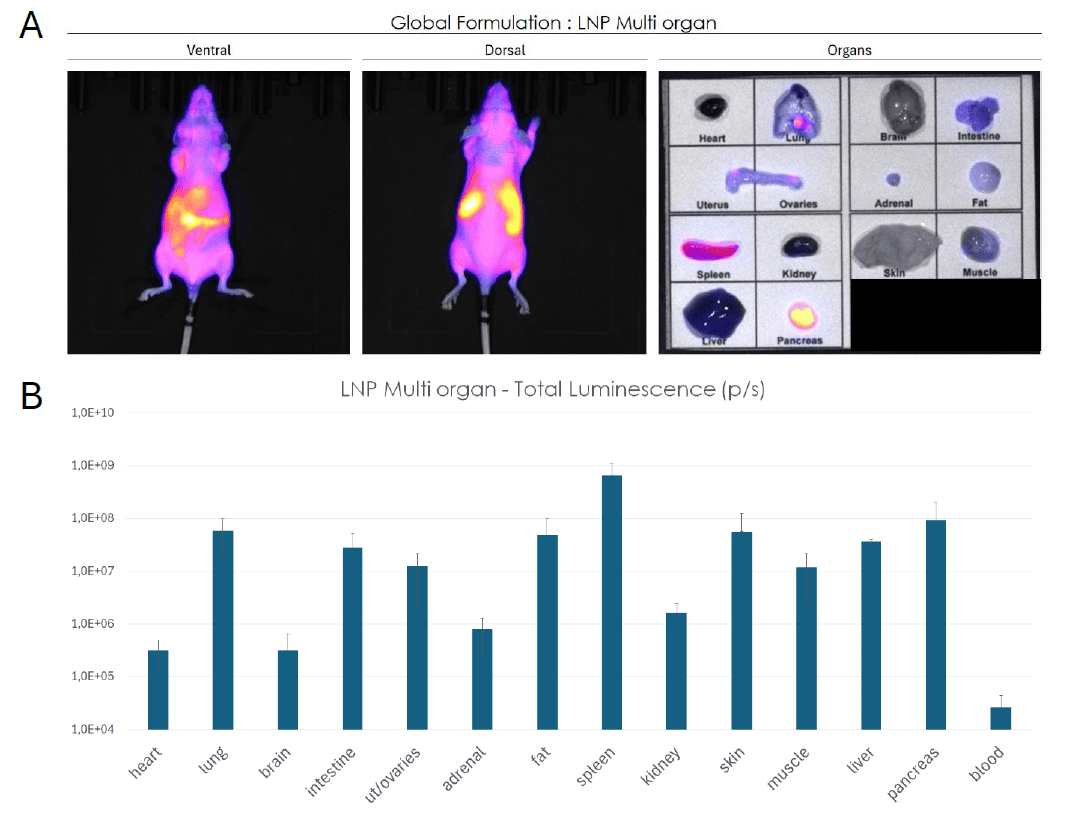
Figure 2: Biodistribution of multi-organ LNP formulation assessed by bioluminescence imaging in mice. (A) Whole-body bioluminescence imaging (ventral and dorsal views) and ex vivo organ imaging following systemic administration of a multi-organ LNP formulation encapsulating F-Luc mRNA. Signal intensity reflects luciferase expression across tissues. (B) Quantification of total luminescence (p/s) per organ, confirming broad tissue tropism with detectable expression across all analyzed organs. Data are presented as mean ± SEM.
b. LNP Liver, a formulation that preferentially targets liver
In contrast to the multi-organ profile, the liver-targeted formulation produced a highly localized signal concentrated in the abdominal region as revealed by the whole body imaging. Ex vivo organ luminescence confirmed preferential luciferase expression in the liver with a secondary contribution from the spleen (Figure 3A). Quantitative analysis demonstrated that liver accounts for more that 70% of total luciferase signal establishing a strong hepatic tropism of this formulation (Figure 3B). Expression in all other organs remained negligible confirming the specificity of the LNP Liver formulation for hepatic target.
Comparison with the reference ionizable lipid SM102 further highlights the superior liver targeting efficiency of this LNP.
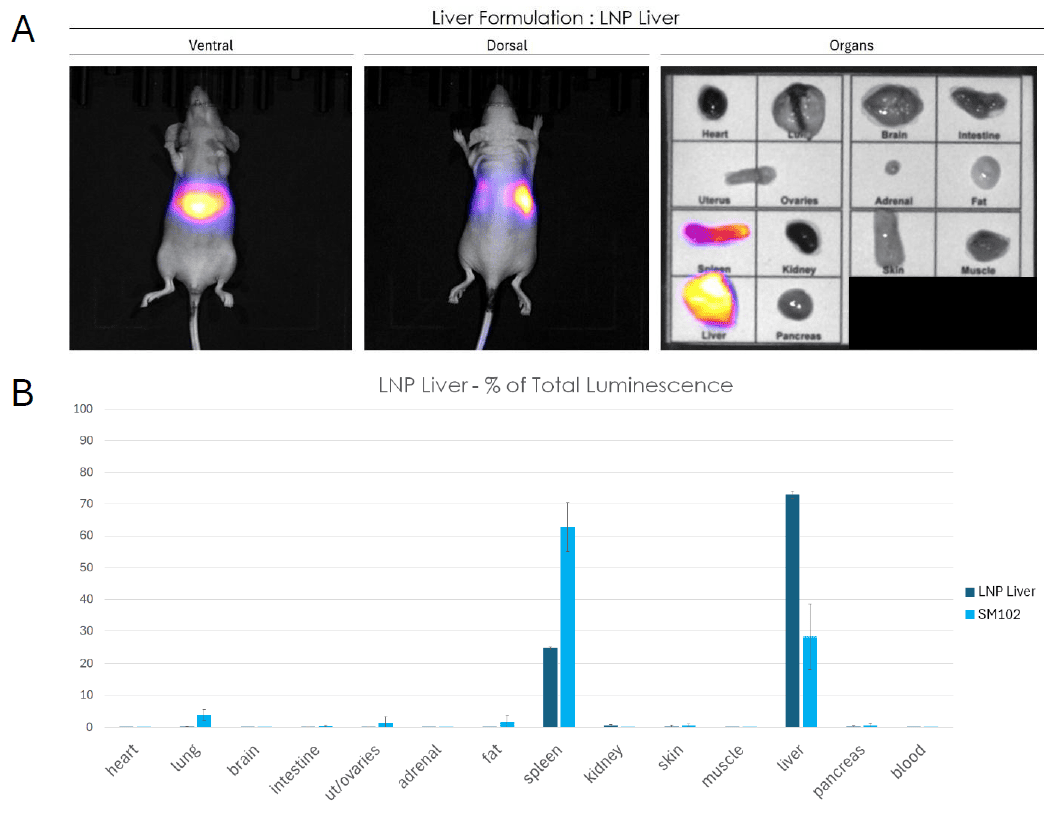
Figure 3: Biodistribution of a liver-targeted LNP formulation assessed by bioluminescence imaging in mice. (A) Whole-body bioluminescence imaging (ventral and dorsal views) and ex vivo organ imaging following systemic administration of LNP Liver formulation encapsulating F-Luc mRNA. Signal intensity is highly concentrated in the abdominal region, with ex vivo imaging confirming dominant expression in the liver and spleen. (B) Quantification of luciferase expression as percentage of total luminescence per organ, comparing LNP Spleen to SM102. The liver accounts for the vast majority of the signal (~73%), demonstrating the strong hepatic tropism of the formulation, with a secondary contribution from the spleen. All other organs show negligible expression. Data are presented as mean ± SEM.
c. LNP spleen, a formulation that preferentially targets spleen
The spleen-targeted formulation achieved a remarkable degree of splenic selectivity. Acquisition of bioluminescence signal demonstrated a peak in splenic region and ex vivo imaging confirmed that luciferase expression was essentially exclusive to the spleen with no detectable signals in the liver or other peripheral organs (Figure 4A). Quantitative data demonstrated that the spleen emitted over than 90% of luminescence signal (Figure 4B).
Compared to SM102, the LNP Spleen formulation displayed dramatically improved splenic tropism with minimal off-target expression.
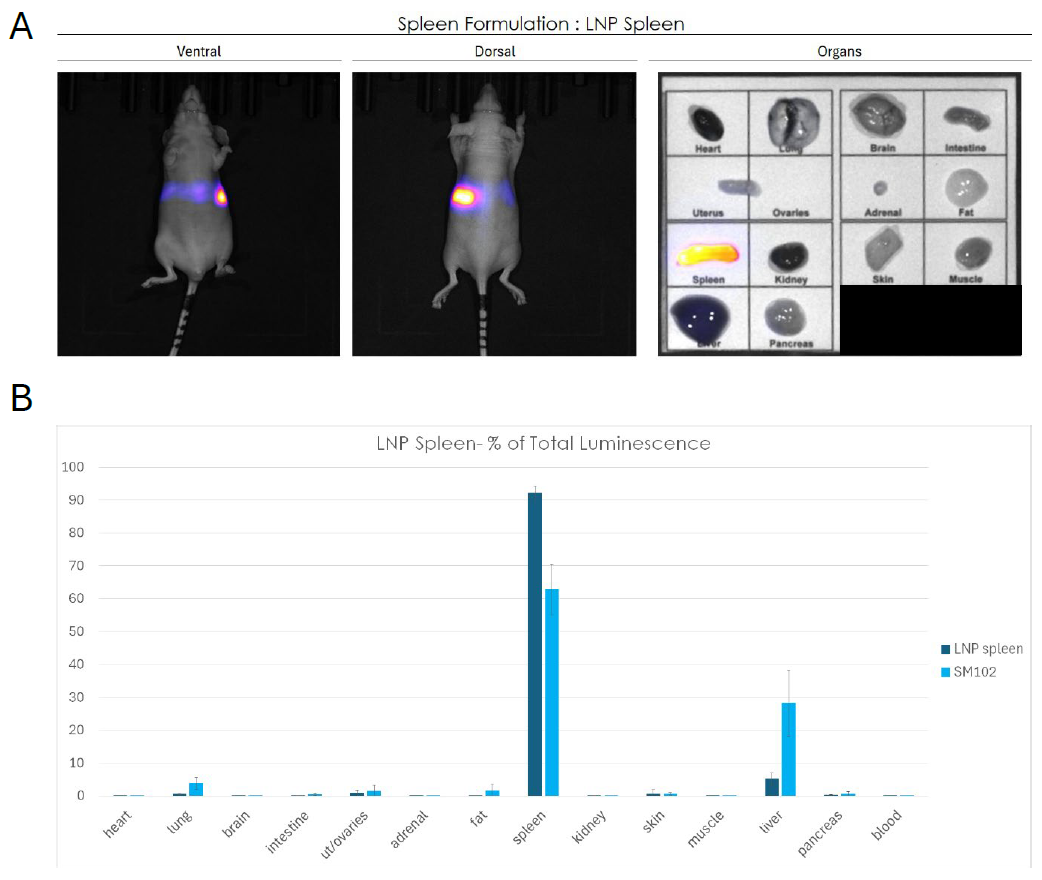
Figure 4: Biodistribution of a spleen-targeted LNP formulation assessed by bioluminescence imaging in mice. (A) Whole-body bioluminescence imaging (ventral and dorsal views) and ex vivo organ imaging following systemic administration of LNP Spleen formulation encapsulating F-Luc mRNA. Signal intensity is localized to the splenic region, with ex vivo imaging confirming exclusive expression in the spleen. (B) Quantification of luciferase expression as percentage of total luminescence per organ, comparing LNP Liver to SM102. The spleen captures over 90% of the total signal with LNP Liver, demonstrating remarkable splenic tropism with minimal off-target expression in all other organs, including the liver. Data are presented as mean ± SEM.
Taken together, these results demonstrate that the ionizable lipids composition is a critital parameter for organ specific delivery of mRNA in vivo. By selecting the appropriate lipid, modulating its concentration and the nature of the associated formulation components, it is possible to redirect LNP biodistribution from whole body expression to precise organs such as liver or spleen.
Ionizable lipid for Immunization
We have evaluated the immunogenicity of different lipid nanoparticles formulation for delivering mRNA encoding antigen in mice. Seven groups of mice (n=5 each) were tested, including untreated controls and treatment groups comparing LNP formulations based on FP105 and FP115 ionizable lipids, with and without mRNA. Using a prime-boost immunization strategy, mice received intramuscular injections of 10 μg mRNA on day 0 and 14. Immune responses were monitored through blood collection on days 28 and 56 for antibody detection, followed by a challenge with purified antigen on day 58. The study concluded on day 63 with tissue collection including thighs for B cell isolation, popliteal and inguinal lymph nodes for immune cell analysis, and sera for comprehensive antibody measurements to assess the relative efficacy of each LNP formulation in generating protective immunity depending on the choice of lipid (figure 5).
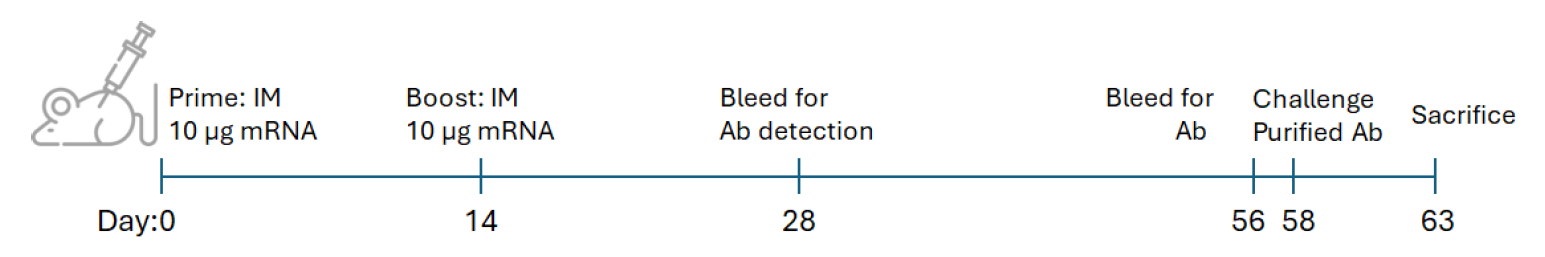
Figure 5: Experimental design for LNP-mRNA immunization in mouse and challenge study. This schematic illustrates the immunization after intramuscular injection of LNP-mRNA and challenge protocol for mice (n=5 per group).
In a parallel experiment we have investigated the immunogenicity of various lipid nanoparticle formulations for delivering mRNA encoding an antigen in mice by subcutaneous injection. Mice received two doses of vaccination using 5 μg mRNA formulated into ionizable lipids-LNP and blood samples were withdrawn each week for 8 weeks.
The results demonstrated that ionizable lipids FP105 and FP115 were 2 interesting candidates for mouse vaccination (Table 4).

Table 4: Choice of ionizable lipid for mRNA-LNP formulation for immunization in mice depending on the site of injection.
Ionizable lipid for gene silencing
LNP-siRNA hold a great potential not only in Research but also show great promises in Therapeutics since many LNP-siRNA based drugs are approved by the FDA such as for example Onpattro for treating Transthyretin Amyloidosis. This is due to the characteristics of these vectors to efficiently deliver the siRNA in various sites depending on LNP generation. Moreover, these vectors are very stable, conferring a prolonged effect (up to 6 months). Chemically speaking, the versatility and the adaptiveness of our LNP mainly comes from their composition with the choice of the ionizable lipid or their decoration with targeting ligands. The creation of any kind of LNP, decorated or not, with our own ionizable lipid or existing ones can be addressed by our LNP platform.
Considering siRNA, we have designed new LNP based on our catalog of ionizable lipid to specifically deliver siRNA aiming at silencing gene expression both in vitro and in vivo. First, a screening was performed using HEK293 cell line expressing GFP to find the best ionizable lipid and LNP composition able to efficiently silence GFP expression. LNP-SS18 and SS23 showed the most important gene silencing 72H after transfection (Figure 6.A). These LNPs were then evaluated in more functional and physiologically relevant model of IL-1 production. PMA primed THP-1 were stimulated during 4 hours with LPS and then transfected with siRNA-IL1 or scramble encapsulated in LNP. 72H after, the production of IL-1 was measured by ELISA and we confirmed that LNP-SS18 and SS23 were ideal to deliver siRNA in vitro (Figure 6.B).
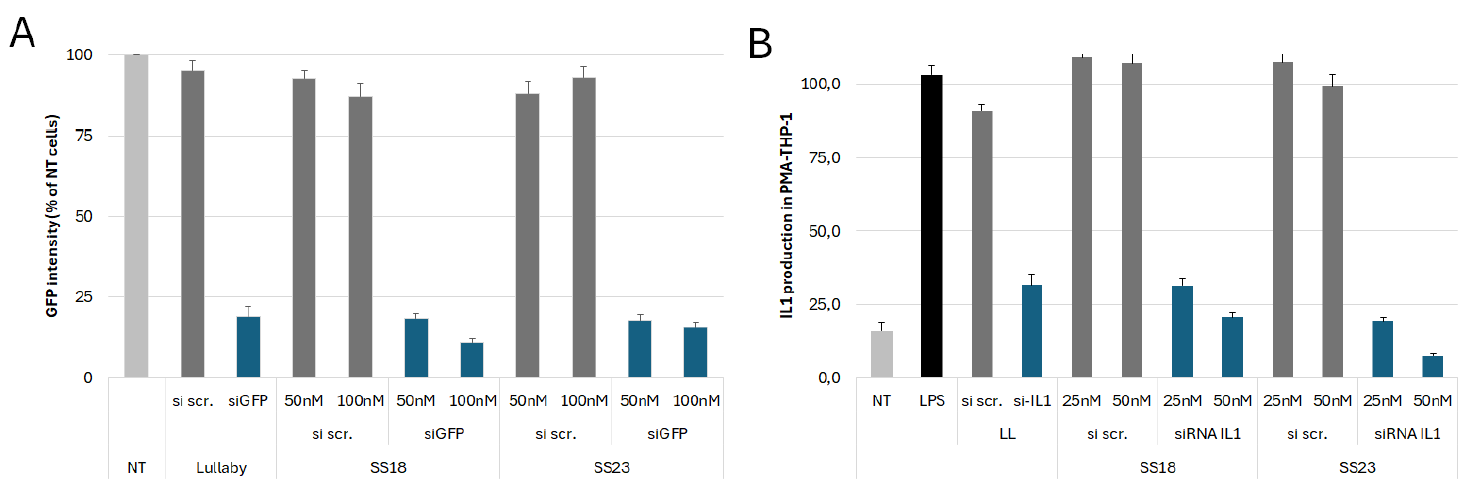
Figure 6: Evaluation of gene silencing efficiency in vitro following siRNA-LNP SS299 and SS23 transfection. (A) HEK293-GFP were transfected with siRNA scrambled (si scr.) or siRNA targeting GFP (siGFP) encapsulated in LNP and GFP intensity was measured 72H after by flow cytometry. (B) Dosage of IL-1 by ELISA in PMA primed THP-1 stimulated with LPS and transfected with si scr. or siRNA targeting IL-1 (siRNA IL1) encapsulated in LNP.
Other ionizable lipids were evaluated in parallel and allowed to silence also gene expression in the two experimental models (data not shown). These results allow us to draw up a table of lipids that can be used to formulate siRNA-LNP aiming at silencing gene expression.

Table 5: Choice of ionizable lipid for siRNA-LNP formulation for gene silencing in HEK293 and macrophages.
siRNA LNP are being tested in vivo in rabbit models of osteoarthritis during the SINPAIN European grant (HORIZON-HLTH-2021-TOOL-06-02). The SINPAIN program aims to develop a pipeline of siRNA-based therapy built on the combination of nanocarriers that will be designed to reach a successful management of inflammation and innervation therapy for the treatment of early (grade 0-1) and later stages (grade 3-4) of knee osteoarthritis. The capacity of the ionizable lipids used to formulate LNP that encapsulates, protects and delivers siRNAs should demonstrate the reduced inflammation and pain, as well as the cartilage regeneration.
Conclusions & future directions
This study demonstrates that ionizable lipid composition is the primary determinant of LNP performance in both in vitro and in vivo settings. With the capacity to modulate composition, charge and size, our LNP platform is able to produce LNPS that can address various zones in the body. As observed, modification of the ionizable lipid can shift organ targeting with LNP that evades liver to target more preferentially the spleen. The data supports a rational design approach where lipid selection is based on target cell type and intended route of administration.
Further optimization of ionizable lipids for cell-type specific delivery remains a priority. Development of lipids with tunable pKa values and biodegradable structures will enhance both efficacy and safety profiles of mRNA therapeutics.
Author: Cédric Sapet, PhD.
References
Cheng, Q., Wei, T., Farbiak, L., Johnson, L. T., Dilliard, S. A., & Siegwart, D. J. (2020). Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR–Cas gene editing. Nature Nanotechnology, 15(4), 313-320.
Melamed JR., Yerneni SS., Arral ML, LoPresti ST… & Whitehead KA. (2023). Ionizable lipid nanoparticles deliver mRNA to pancreatic β cells via macrophage-mediated gene transfer. Sci Adv. Jan 27;9(4):eade1444.
Patel, S., Ashwanikumar, N., Robinson, E., Xia, Y., Mihai, C., Griffith, J. P., ... & Sahay, G. (2020). Naturally-occurring cholesterol analogues in lipid nanoparticles induce polymorphic shape and enhance intracellular delivery of mRNA. Nature Communications, 11(1), 983.
Dilliard, S. A., Cheng, Q., & Siegwart, D. J. (2021). On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticles. Proceedings of the National Academy of Sciences, 118(52), e2109256118.
Carrasco MJ., Alishetty S., Alameh MG., …. & Buschmann MD. (2021). Ionization and structural properties of mRNA lipid nanoparticles influence expression in intramuscular and intravascular administration. Commun Biol., Aug 11;4(1):956.
Lee AM., Cheng Q., Yu X., Liu S., Jonhson LT., & Siegwart DJ. (2021). A Systematic Study of Unsaturation in Lipid Nanoparticles Leads to Improved mRNA Transfection In Vivo. Angew Chem Int Ed Engl. Mar 8;60(11):5848-5853.