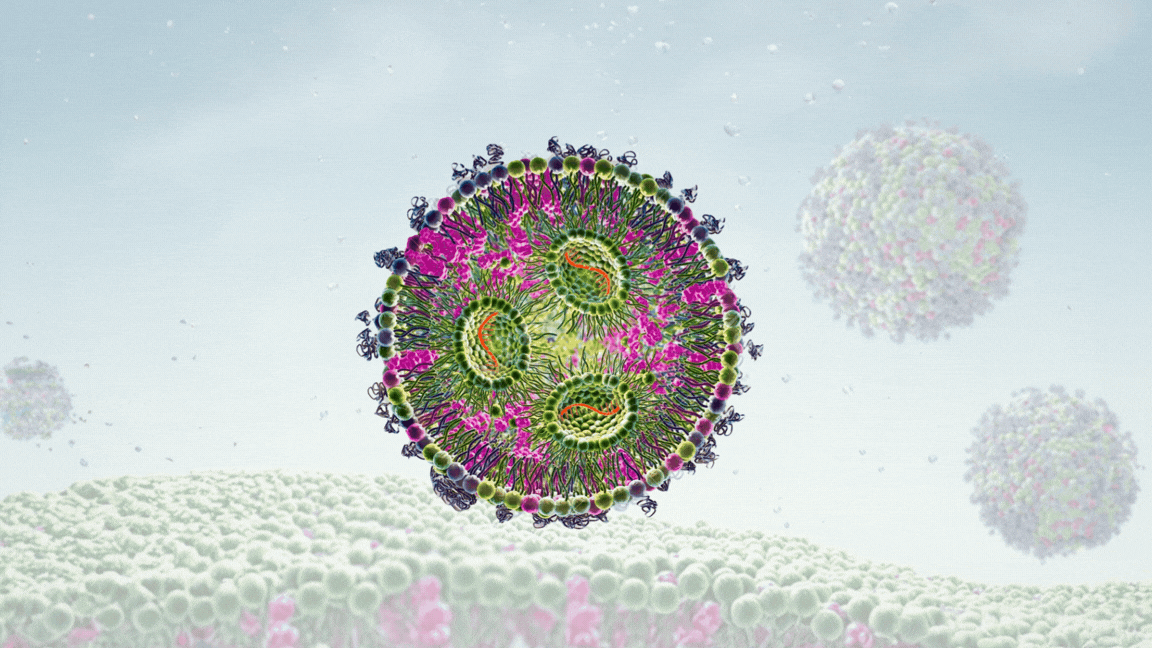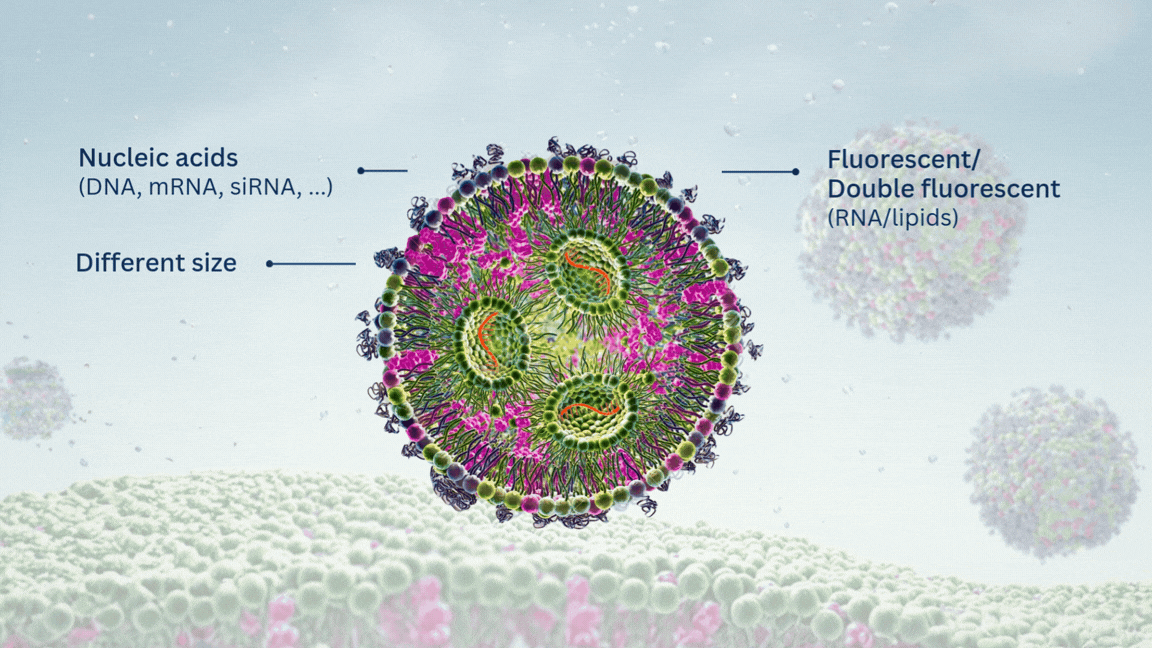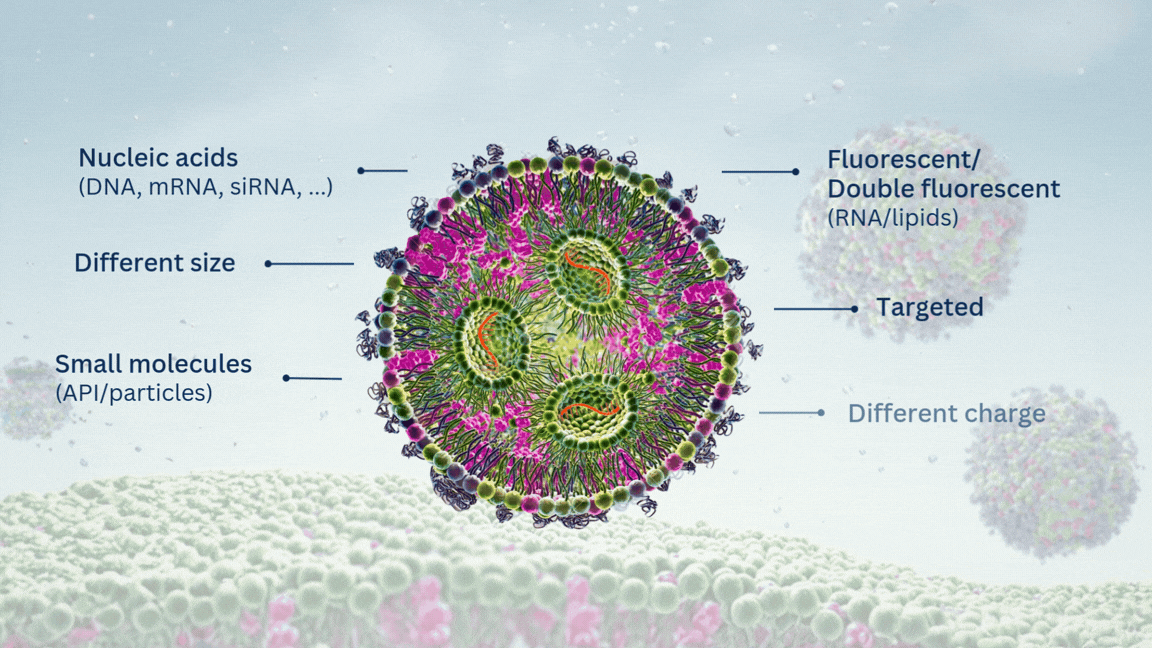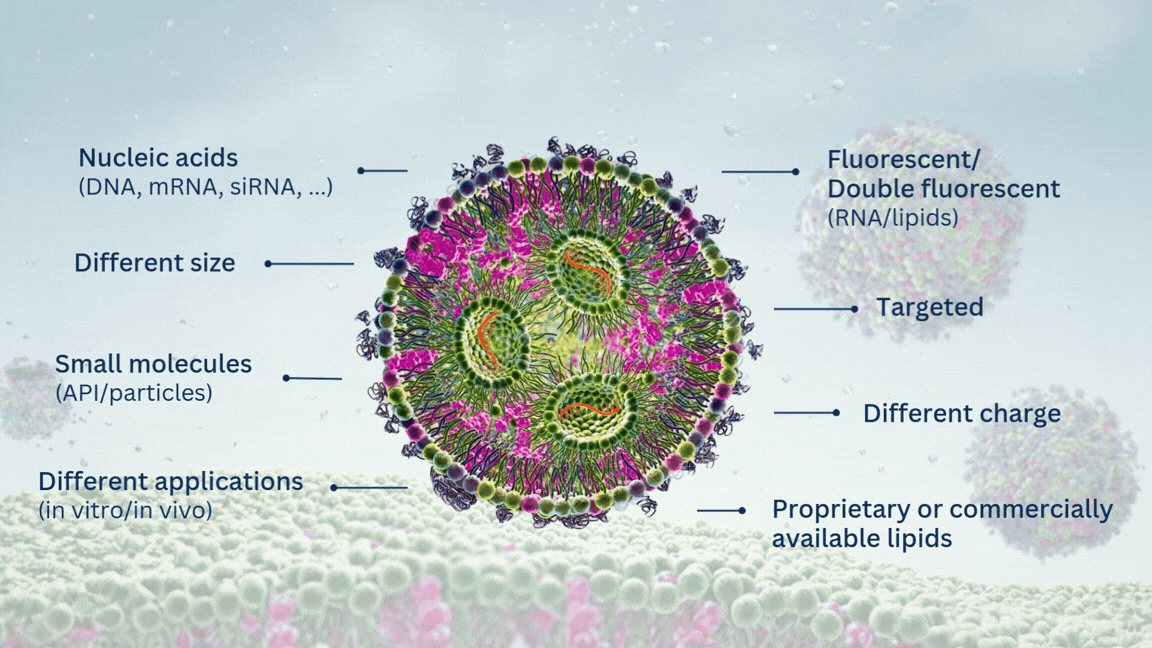
Highly Potent Non-Viral Drug Delivery Nanosystems
OZ Biosciences has developed a Microfluidics Platform for the reproducible development of safe & potent drug delivery vehicles for pharmaceutical applications.
Our platform can produce custom LNP according to your needs and preferences.
Custom LNP service workflow
Our team is dedicated in providing you with high quality LNP that will meet your project needs. You can provide your own molecule of interest to encapsulate or we can make a custom mRNA for you.
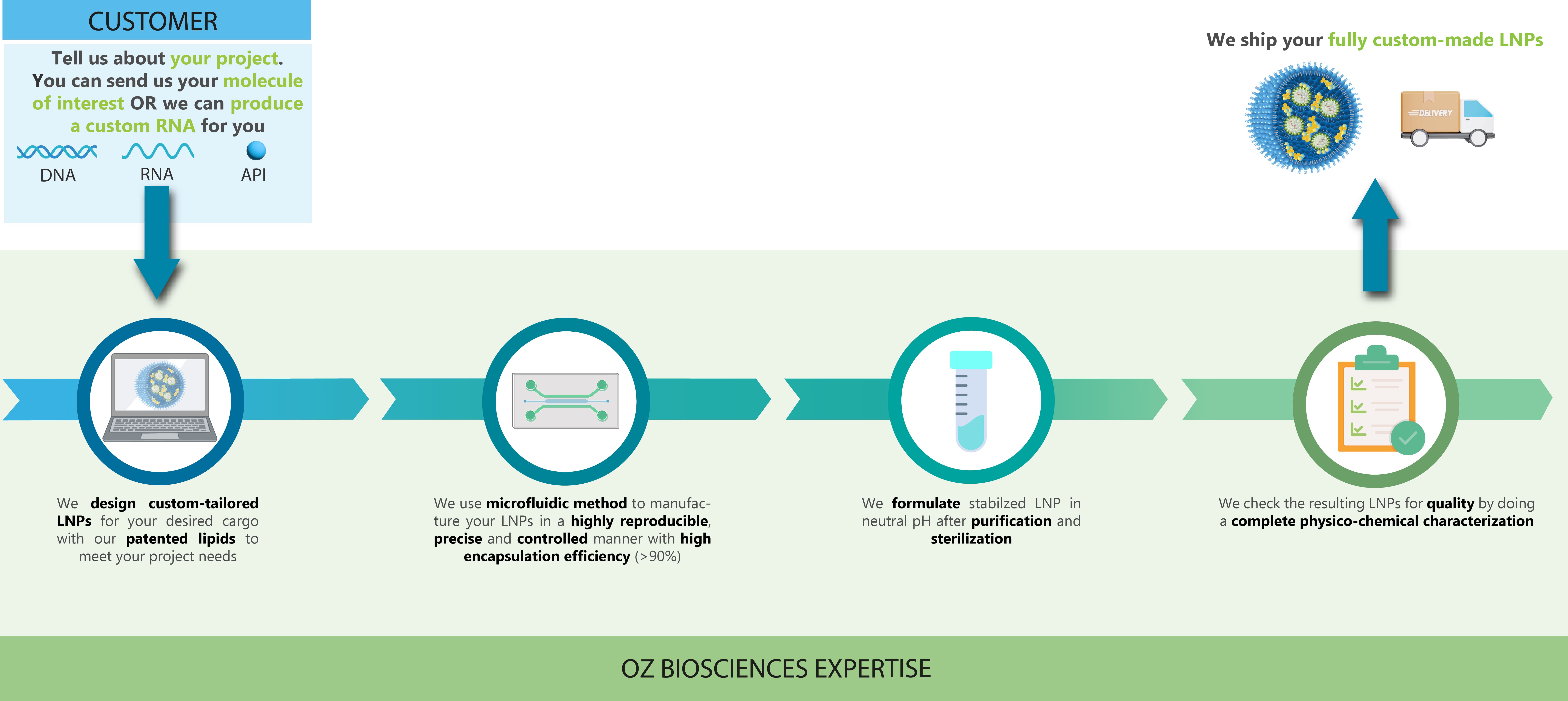
We can support every stage of your mRNA-LNP production, from mRNA synthesis to LNP formulation development, manufacturing and fill & finish.
For any of RNA, DNA or APIs encapsulation, you can provide us with your molecule of interest and we will formulate it into LNPs.
Our expert product support team is at your service for your special requests - you are invited to reach out to our experts to discuss your needs.
LNP Performance Data by Application
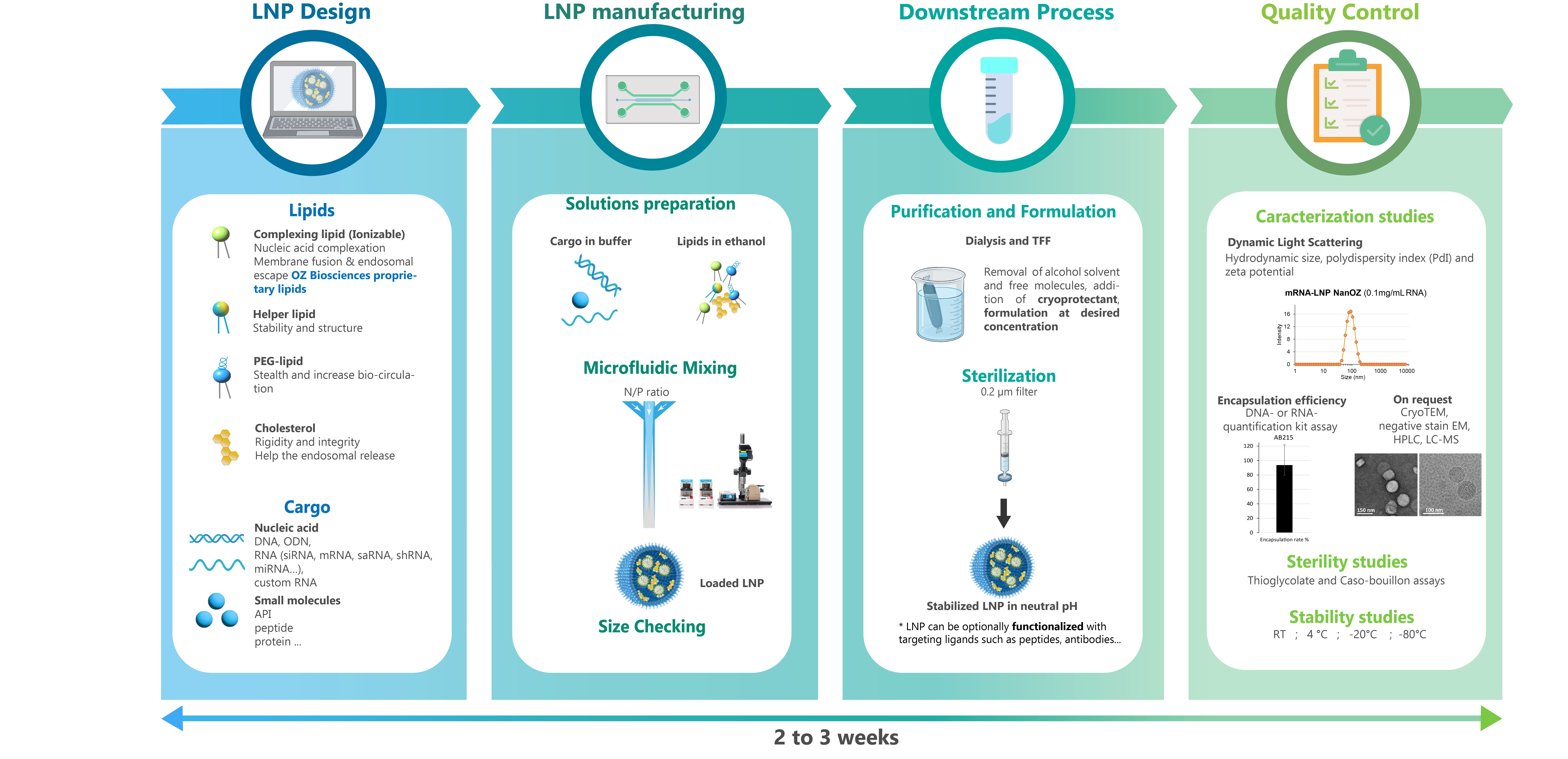
NanOZ-LNPs are formulated through pressure-driven controlled flow microfluidics systems. Once collected and purified, NanOZ-LNPs physico-chemical properties are fully characterized in terms of size distribution, charge surface, structure integrity, encapsulation efficiency and stability.
The NanOZ-LNP/mRNA are monodisperse nanoparticles of spherical morphology with size usually ranging between 80-150nm and PDI<0.2. Once encapsulated, the integrity of the mRNA is verified by gel electrophoresis.
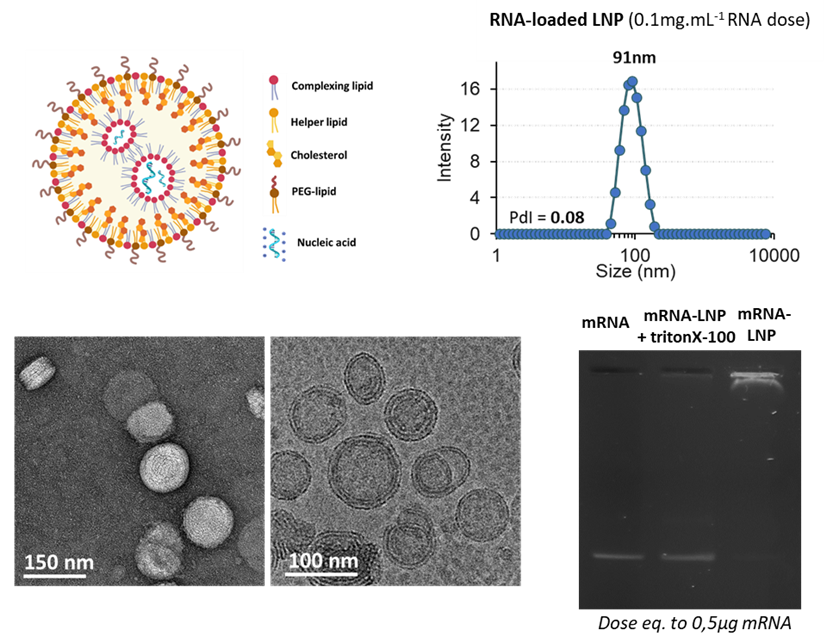
Figure 1: Physico-chemical characterizations of NanOZ-LNP/mRNA: Size distribution and Polydispersity Index measurements by DLS, morphology and monodispersity observed by colour negative SEM and CryoTEM micrographies and mRNA integrity after formulation inside LNP monitored by agarose gel electrophoresis at mRNA dose equals to 0.5µg. Data Source: The results obtained by OZB, Marseille.
To evaluate the organ-targeting capabilities of our ionizable lipid library, mRNA encoding Firefly Luciferase (F-Luc mRNA #MRNA16-100) was encapsulated in lipid nanoparticles (LNPs) formulated with a panel of proprietary ionizable lipids. All formulations were administrated intravenously to mice, and luciferase expression was monitored 6 hours post-injection by bioluminescence imaging following intravenous injection of D-Luciferin. Both whole-body imaging (ventral and dorsal views) and ex vivo organ imaging were performed to accurately map the biodistribution of each formulation at the tissue level.
The experimental design revealed that the choice of ionizable lipid, as well as its concentration and the nature of the associated lipid components, is a primary determinant of LNP tropism following systemic delivery.
Depending on the formulation of the LNP, mRNA delivery could be directed preferentially to a single organ, distributed broadly across multiple tissues or finely tuned by adjusting the lipid composition.
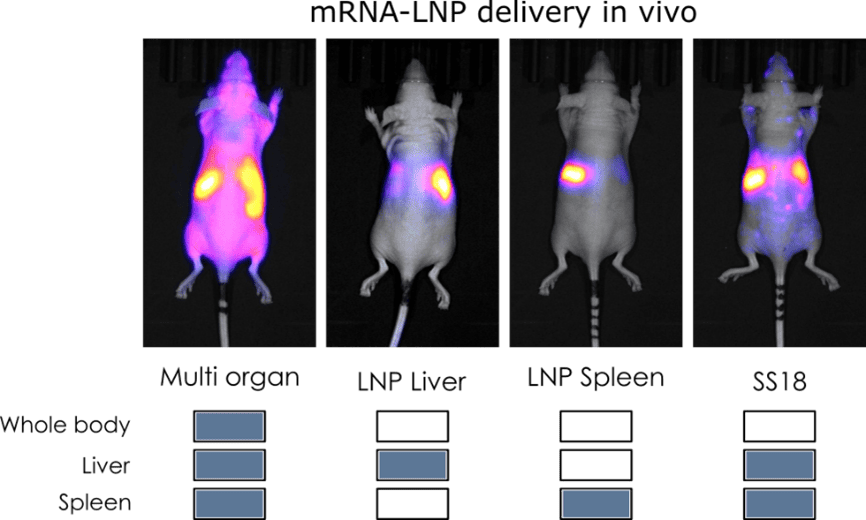
Taken together, these results demonstrate that the ionizable lipids composition is a critital parameter for organ specific delivery of mRNA in vivo. By selecting the appropriate lipid, modulating its concentration and the nature of the associated formulation components, it is possible to redirect LNP biodistribution from whole body expression to precise organs such as liver or spleen.
The comparison between mRNA-based vaccines versus subunit vaccine for immune efficacy were evaluated in C57BL6J mice using the ovalbumin model. Prime boost (D0-14) immunizations were evaluated with OVA antigen delivered by mRNA-based LNPs, where two different formulations were tested corresponding to LNP1 and LNP2, or by recombinant protein (alone or adjuvanted). To characterize the immuno-phenotyping of the mice the sera were collected at D0, D14, D21 and D28 and splenocytes were collected at D28 and analysed by ELISA, Elispot and Multiplex. The mice were immunized by s.c. route with 10µg OVA subunit (SU) or 10µg mRNA doses (100µL per injection).
RNA vaccine LNP formulations are efficient at priming a strong Th1 humoral response equivalent to the best TH1 adjuvants such as QS-21 upon sub-cutaneous prime boost injection (D0-14) in C57BL6J mice. In addition, no significative signal was observed when the OVA antigen is directly administrated. It is noteworthy, the necessity of two injections (boost at D14) to obtain a long-lasting immune response.
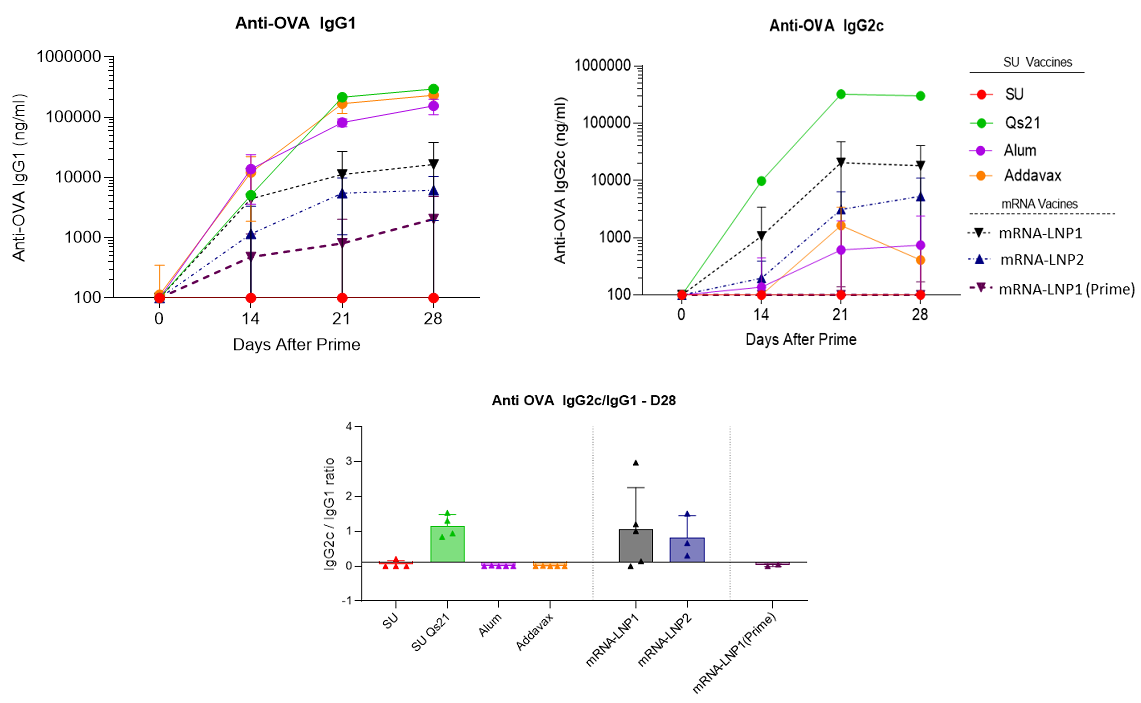
Figure 3: Anti-OVA IgG production and Th1 humoral biomarker to OVA subunit vaccine (10µg) in absence or presence of vaccines adjuvants (QS21 (5µg), Alum (Aluminum Hydroxide gel)1X or addaVax 1X) and to mRNA(OVA)-LNPs (mRNA dose equals to 10µg) upon sub-cutaneous prime boost injection (D0-14) in C57BL6J mice. Data Source: The results were acheived by OZ Biosciences in collaboration with Dr. Antoine Tanne, SaponiQx, Lexington, MA, USA
Optimization of LNPs by Modulating OZB-Property ionizable lipids for Enhanced mRNA Transfection in Various Cell Lines (Dendritic, macrophage, monocyte, T cells).
After extensive R&D efforts in lipid synthesis, five ionizable lipids were selected for screening the transfection efficiency of LNP-mRNA formulations. In this study, two mRNA reporter genes, enhanced green fluorescent protein (eGFP) and firefly luciferase (Fluc), were used. The selected LNPs were tested on the following cell lines: DC2.4, RAW 264.7, J774, THP-1, Jurkat, MOLT-4. Transfection was performed using LNP-mRNA at doses of 0.1, 0.5, 1, 2, and 5 μg of mRNA, with Luciferase and GFP expression assessed 24 hours post-transfection. The LNP formulated with the ionizable lipid FP105 corresponds to our catalogue product NanOZ. It is important to note that the direct comparison or translation of findings from in vitro to in vivo conditions is highly complex. This is due to fundamental differences in physiological environments, including factors such as biodistribution, cellular uptake, metabolic stability, and immune system interactions, which cannot be fully replicated in vitro.
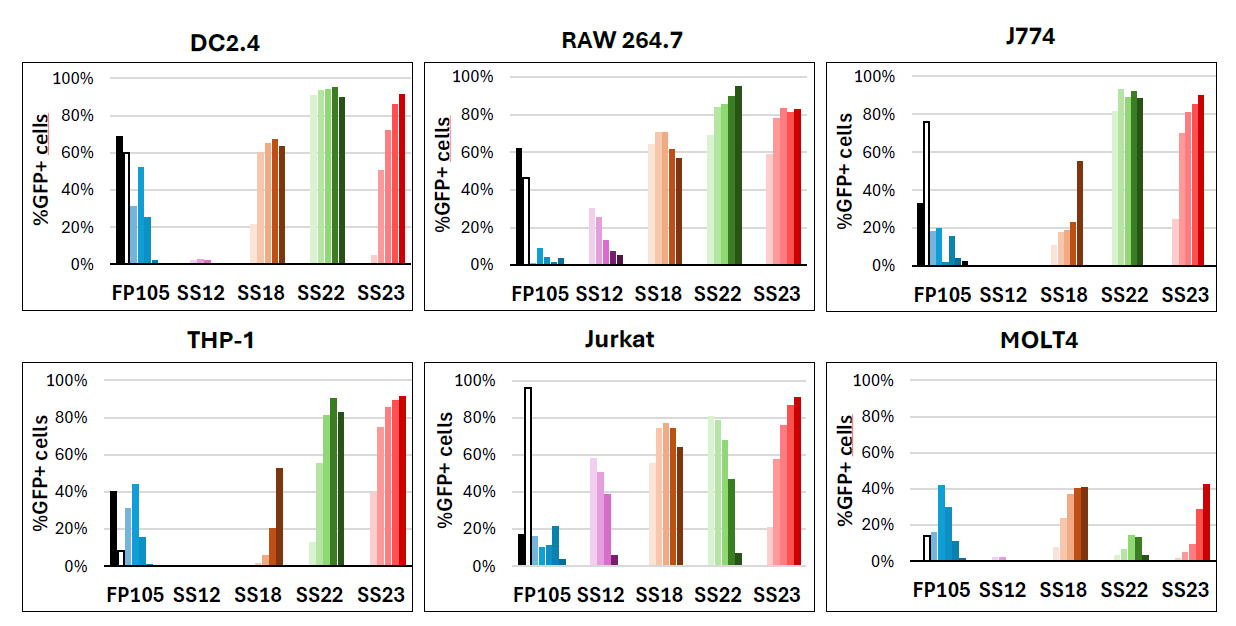
Figure 4: Flow cytometry (FACS) analysis shows the transfection efficiency expressed as the percentage of GFP-positive cells using doses ranging from 0.1, 0.5, 1, 2, and 5 μg of mRNA per well of 24-well plates in various cell lines (DC2.4, RAW 264.7, J774, THP-1, Jurkat, MOLT-4). Transfection efficiency is represented as the percentage of cells expressing GFP. The black bar represents traditional lipoplex (RmesFect), while the white bar represents LNP formulated with the well described and FDA-approved ionizable lipid SM102.
LNP-siRNA hold a great potential not only in Research but also show great promises in Therapeutics since many LNP-siRNA based drugs are approved by the FDA such as for example Onpattro for treating Transthyretin Amyloidosis. This is due to the characteristics of these vectors to efficiently deliver the siRNA in various sites depending on LNP generation. Moreover, these vectors are very stable, conferring a prolonged effect (up to 6 months). Chemically speaking, the versatility and the adaptiveness of our LNP mainly comes from their composition with the choice of the ionizable lipid or their decoration with targeting ligands. The creation of any kind of LNP, decorated or not, with our own ionizable lipid or existing ones can be addressed by our LNP platform.
Considering siRNA, we have designed new LNP based on our catalog of ionizable lipid to specifically deliver siRNA aiming at silencing gene expression both in vitro and in vivo. First, a screening was performed using HEK293 cell line expressing GFP to find the best ionizable lipid and LNP composition able to efficiently silence GFP expression. LNP-SS18 and SS23 showed the most important gene silencing 72H after transfection (Figure 6.A). These LNPs were then evaluated in more functional and physiologically relevant model of IL-1 production. PMA primed THP-1 were stimulated during 4 hours with LPS and then transfected with siRNA-IL1 or scramble encapsulated in LNP. 72H after, the production of IL-1 was measured by ELISA and we confirmed that LNP-SS18 and SS23 were ideal to deliver siRNA in vitro (Figure 6.B).
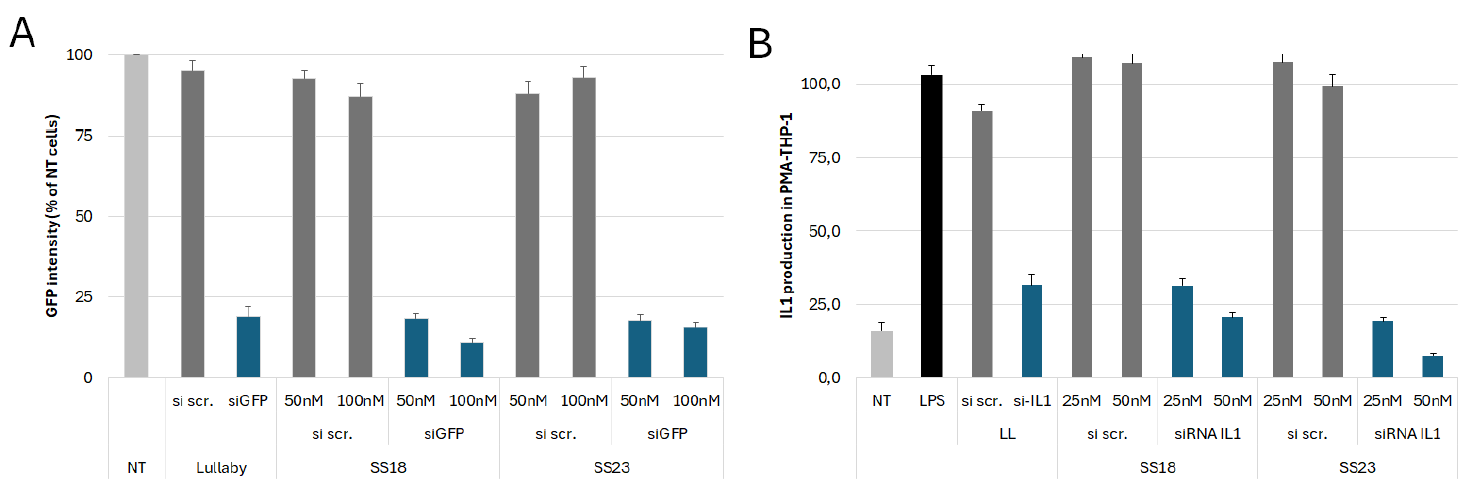
Figure 6: Evaluation of gene silencing efficiency in vitro following siRNA-LNP SS299 and SS23 transfection. (A) HEK293-GFP were transfected with siRNA scrambled (si scr.) or siRNA targeting GFP (siGFP) encapsulated in LNP and GFP intensity was measured 72H after by flow cytometry. (B) Dosage of IL-1 by ELISA in PMA primed THP-1 stimulated with LPS and transfected with si scr. or siRNA targeting IL-1 (siRNA IL1) encapsulated in LNP.
Other ionizable lipids were evaluated in parallel and allowed to silence also gene expression in the two experimental models (data not shown). These results allow us to draw up a table of lipids that can be used to formulate siRNA-LNP aiming at silencing gene expression.

Table 5: Choice of ionizable lipid for siRNA-LNP formulation for gene silencing in HEK293 and macrophages.
siRNA LNP are being tested in vivo in rabbit models of osteoarthritis during the SINPAIN European grant (HORIZON-HLTH-2021-TOOL-06-02). The SINPAIN program aims to develop a pipeline of siRNA-based therapy built on the combination of nanocarriers that will be designed to reach a successful management of inflammation and innervation therapy for the treatment of early (grade 0-1) and later stages (grade 3-4) of knee osteoarthritis. The capacity of the ionizable lipids used to formulate LNP that encapsulates, protects and delivers siRNAs should demonstrate the reduced inflammation and pain, as well as the cartilage regeneration.
Technical Resources
- LNP Stability Note - Download
- White Paper : Ionizable lipids for mRNA-LNP and siRNA-LNP - Read
- LNP F.A.Q - Read
Custom LNP F.A.Q
You can easily request a quote by filling in our dedicated form available here. If you need further guidance or wish to discuss your project and specific requirements with our technical team, feel free to contact us — we will be happy to arrange a meeting at your convenience.
Once the mRNA or nucleic acid cargo is ready, our LNP production process typically takes 2 to 3 weeks: approximately one week for production and one week for quality control. If custom mRNA synthesis is also required, please allow an additional 8 weeks for mRNA production prior to LNP formulation.
The minimum order quantity for our custom LNP formulation service is 0.5 mL. For specific requirements outside this range, please contact us to discuss available options.
Our custom LNP formulation service supports encapsulation of a wide range of molecules, including mRNA, siRNA, saRNA, DNA, ASO/ODN, and other molecules of interest. Regarding the origin of the cargo, we offer three options:
- OZB catalog mRNA: choose from our existing catalog of ready-to-use mRNAs (e.g. GFP, Luciferase, OVA, Spike).
- OZB custom mRNA: we produce a custom mRNA from your sequence of interest.
- Customer-supplied molecule: you provide your own molecule, which we will encapsulate into our LNP system.
Simply indicate your preferences when filling in our dedicated custom LNP formulation quote form and our team will get back to you with a tailored proposal.
Yes. As part of our custom service, we can work with alternative ionizable lipids or entirely different LNP compositions to meet your specific delivery requirements; whether for targeted delivery, specialized routes of administration, or unique cargo types. If you are able to provide your own lipids, we can incorporate them into our formulation process. Simply specify this when filling in our custom LNP formulation quote form and we will proceed accordingly.
Yes, the final concentration can be modulated upon request. However, achievable concentrations may vary depending on the nature and amount of the encapsulated molecule. Please specify your concentration requirements when submitting your order or quote request.
Yes. As an option, our LNPs can be functionalized with targeting ligands such as peptides or antibodies, enabling active targeting strategies for specific cell types or tissues. Please contact us or fill in our custom LNP formulation quote form to discuss the feasibility for your application.
Each LNP batch undergoes a comprehensive quality control workflow including particle size and PDI measurement by DLS, zeta potential analysis, encapsulation efficiency determination by fluorescent assay, and mRNA concentration measurement. The sterility is also assessed by incubating the LNPs in culture medium for 14 days. For superior grade products, endotoxin testing and cholesterol content quantification are additionally performed.
Please refer to our LNP quoting tool to select the QC level that best fits your project requirements.
Discover our other custom services

Custom mRNA Synthesis Service
OZ Biosciences offers mRNAs that mimic fully processed mature mRNAs.
These mRNAs are stabilized with 5’ Cap 1 structure and 3’ poly(A) tail and are...

Custom saRNA Synthesis Service
Self-amplifying RNAs (saRNAs) also called “Replicons” are the next generation of RNA vaccines.
Their advantage over conventional mRNA vaccine platforms relies on the viral replication machinery,...