BLOG > Publications & Citations > Cytokine-fusion scaffold generates CAR T cells enriched in long-lived TSCM
Rethinking CAR T Cell Manufacturing: Cytokine Fusion Scaffold Unlocks Long-Lived T Memory Stem Cells
CAR T cell therapy has transformed oncology with the approval of five therapies for treatment of leukemia, lymphoma and multiple myeloma and is currently advancing in the field of infectious diseases. However, one fundamental limitation persists: longevity. The standard manufacturing workflow which relies on anti-CD3/CD28 (aCD3/28) stimulation to activate T cells before lentiviral transduction, efficiently expands cells but drives them towards terminal differentiation. The resulting CAR T cells are potent short-term killers, but they tend to exhaust rapidly in vivo, limiting long-term disease control.
In this remarkable study published in Science Advances, Erin Cole et al. from Albert Einstein College of Medicine & HCW Biologics, addressed this fundamental problem by replacing aCD3/28 with HCW9206; a unique soluble cytokine-fusion scaffold linking IL-7, an IL-15/IL-15Rα sushi domain complex (IL-15SA), and IL-21 and the results are striking.
Key results
This study demonstrates that replacing conventional aCD3/28 activation with HCW9206 cytokine scaffold generates CAR T cells with a ~14 fold enrichment in T memory stem cells (TSCM) for anti-HIV duoCAR and ~12-fold for anti-CD19 CAR without compromising effector function. Superior in vivo persistence and tumor control upon rechallenge was also described. These results were built on two key contributions from LentiBlast Premium. (1) First, used during the transduction of primary human T cells in both manufacturing arms, it delivered consistently high CAR expression (60-80% CAR+ CD8+ T cells) across multiple donors and both CAR constructs. This ensured that the dramatic phenotype differences observed between the two groups could be attributed entirely to the activation method, and not to variability in gene delivery. (2) Second, used at 2 µL/mL to generate NALM-6-Luc reporter line, LentiBlast Premium enabled real-time IVIS tracking of tumor burden in vivo. The generation of bioluminescent NALM-6-Luc tumor line powered the in vivo rechallenge model that allowed to reveal the most striking result from the study: complete suppression of leukemia regrowth upon rechallenge in all mice treated with HCW9206-generated CAR T cells.
Methodology: comparing two CAR T cell manufacturing workflows applied to both anti-HIV duoCAR and anti-CD19 CAR construct
- Standard arm: CD4+ and CD8+ T cells were activated with aCD3 and aCD28 antibodies in the presence of IL-2 for 3 days, then transduced with CAR-encoding lentivirus in presence of LentiBlast Premium.
- HCW9206 arm: T cells were treated with 100 nM HCW9206 for 7 days before LentiBlast Premium enhanced lentiviral transduction without TCR stimulation.
- Transduction in presence of LentiBlast Premium was performed by spinoculation (2500 rpm x 1139xg x 60 min) at an MOI of 40-50. Efficiency was evaluated by flow cytometry 5-10 days post transduction.
- In vivo leukemia model: bioluminescent NALM-6-Luc reporter line was engineered by transducing NALM-6 cells with a Luciferase-tdTomato lentiviral vector in presence of LentiBlast Premium (2µL/mL).
- Post transduction characterization included: multiparametric flow cytometry, progeny and proliferation assays, intracellular cytokine staining, RNA sequencing, In vitro HIV suppression assay or in vivo experiments.
LentiBlast Premium in this study
LentiBlast Premium (LBPX500, OZ Biosciences) was used at two critical and distinct steps of the experimental workflow, illustrating the versatility across both primary T cell transduction and cell line engineering.
Step 1: CAR T cell lentiviral transduction.
Following either aCD3/28 activation or HCW9206 treatment, highly purified primary CD4+ and CD8+ T cells were transduced with anti-HIV duoCAR or anti-CD19 CAR lentiviral vectors in the presence of LentiBlast Premium. Transduction were performed by spinoculation and cells were incubated 5 to 10 days before evaluation of the experiment by flow cytometry.
As shown in figure 2B, the full HCW9206 workflow consists of a 7-day cytokine pre-treatment followed by lentiviral transduction in the presence of LentiBlast. Protocol was free of TCR stimulation.
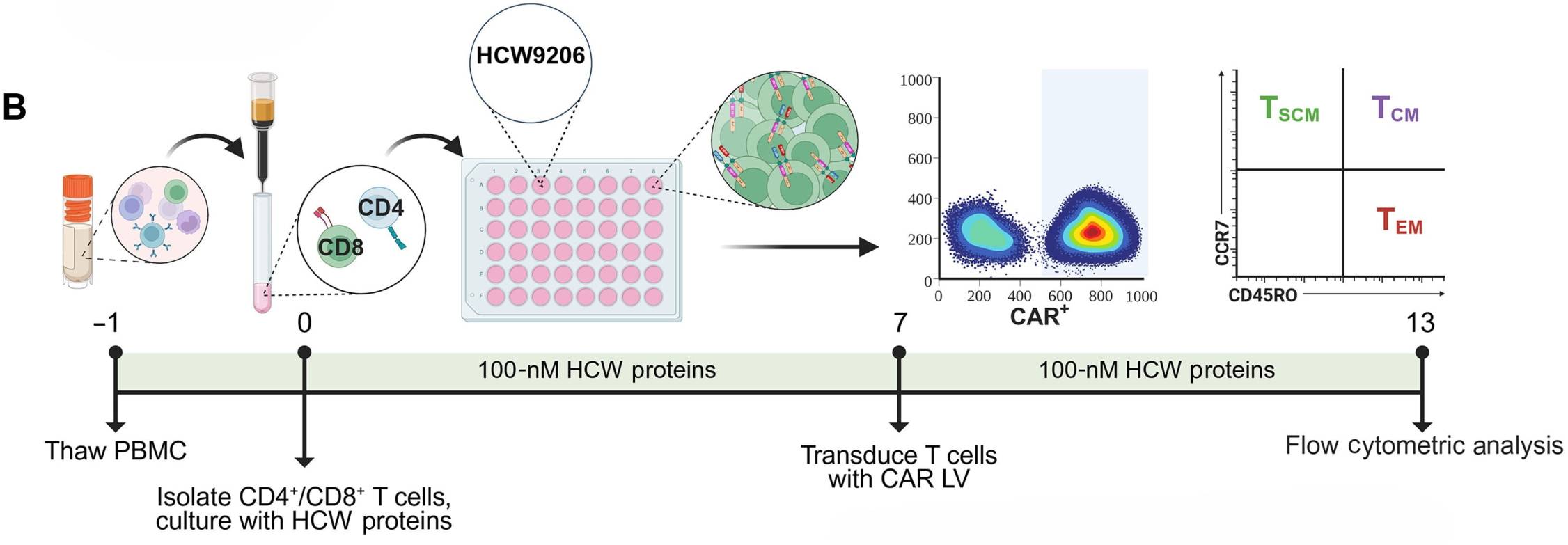
Figure 2B: protocol for HCW9206-generated anti-HIV duoCAR T cell product. T cells were treated with HCW9206 (100 nM) for 7 days or activated with aCD3/28 for 3 days before transduction with CAR-encoding lentivirus in presence of LentiBlast Premium. (Adapted from Cole et al., Sci Adv. 12, eaec2632 (2026).
The key results are demonstrated in Figure 2C and D, that show that comparable duoCAR expression levels (detected by m36.4 and mD1.22 surface markers) were achieved in both HCW9206 and aCD3/28 arms, reaching 60-80%. Significantly, as shown in Figure 2E, while transduction efficiency was equivalent between the two conditions, the fraction of duoCAR T cells displaying a TSCM phenotype was ~14 fold higher in the HCW9206 arm demonstrating that LentiBlast Premium supported efficient gene delivery under both conditions allowing the full phenotypic advantage of HCW9206 to emerge.
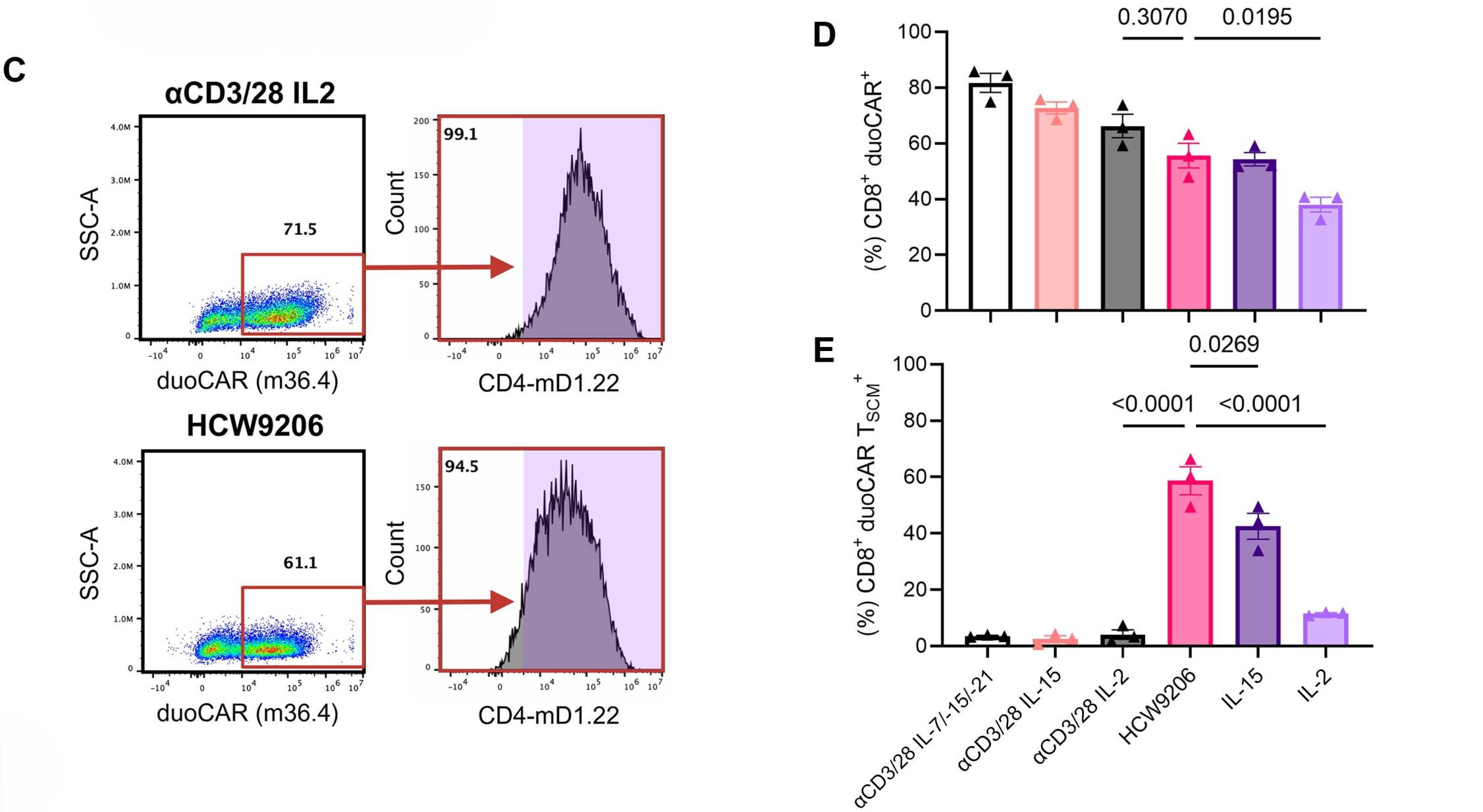
Figure 2C, D, E: LentiBlast induced Lentiviral transduction efficiency and TSCM cell enrichment in HCW9206-generated duoCAR T cells. (C) Representative fluorescence-activated cell sorting (FACS) dot and histogram plots [Y axis = SSC-A (side scatter area)] showing CD8+ T cell expression of m36.4 and mD1.22 duoCAR following treatment with HCW9206 (100 nM) or αCD3/28 + IL-2 (100 U/ml). (D) DuoCAR expression in age-matched donor CD8+ T cells after αCD3/28 activation or HCW9206 or soluble cytokine treatment (N = 3). Gated on lymphocytes, single cells, CD8+, and m36.4+ (N = 3). (E) Frequency of CD8+ duoCAR TSCM cells following αCD3/28 activation or cytokine treatment. (Adapted from Cole et al., Sci Adv. 12, eaec2632 (2026).
The same results were obtained with the anti-CD19 construct. As shown in Figure 3B, LentiBlast Premium enabled comparable CD19 CAR expression across all conditions tested. Figure 3C further reveals the striking phenotypic advantage of HCW9206: while transduction efficiency was equivalent between the two conditions, CD19 CAR T cells generated with HCW9206 and LentiBlast Premium were massively enriched in TSCM cell markers compared to aCD3/28 generated cells, which displayed a predominantly more differentiated phenotype. This enrichment is quantified in Figure 3D, showing a ~12 fold higher frequency of CD19 CAR TSCM cells in the HCW9206 arm. Together, figures 3D, C and D illustrate that LentiBlast Premium delivered consistent gene transfer across both protocols, allowing to demonstrate full TSCM enrichment driven by HCW9206.
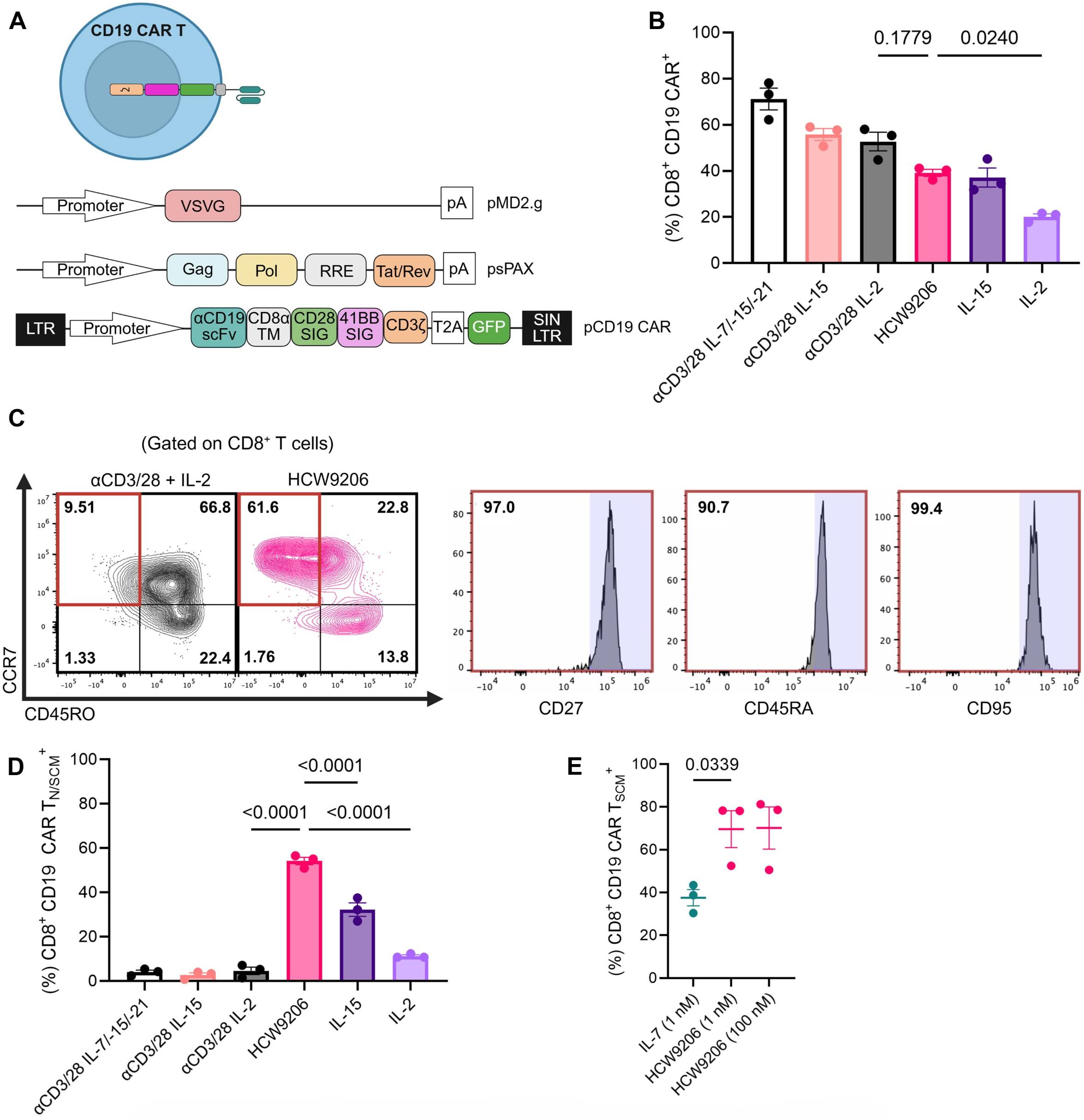
Figure 3A, B, C, D & E: CD19 CART T cell transduction and TSCM enrichment. LentiBlast Premium delivered comparable CD19 CAR expression across all conditions. (B) CCW9206 generated cells showed a ~12 fold enrichment in TSCM cells confirmed by FACS (C) and quantification (D). (E) Dose dependent superiority of HCW9206 scaffold over IL-7 alone. (Adapted from Cole et al., Sci Adv. 12, eaec2632 (2026).
Step 2: Engineering the NALM-6-Luc reporter cell line.
The in vivo leukemia rechallenge model presented in Figure 6 was entirely dependant on the bioluminescent NALM-6-Luc reporter cell line generated using LentiBlast Premium at 2 µl/mL. As illustrated in panel B and C, real-time IVIS imaging tracked tumor burden over 43 days revealing complete suppression of NALM-6-Luc expansion upon rechallenge in all mice treated with HCW9206-generated CAR T cells that were transduced with LentiBlast Premium, versus progressive tumor dissemination in 5 out of 6 mice in the aCD3/28 group. Panel D further shows that only HCW9206-generated CAR T cells displayed a robust recall proliferative response in peripheral blood upon rechallenge, while aCD3/28-generated cells remained barely detectable. Altogether the results demonstrate the long-term functional persistence conferred by the HCW9206 manufacturing protocol.
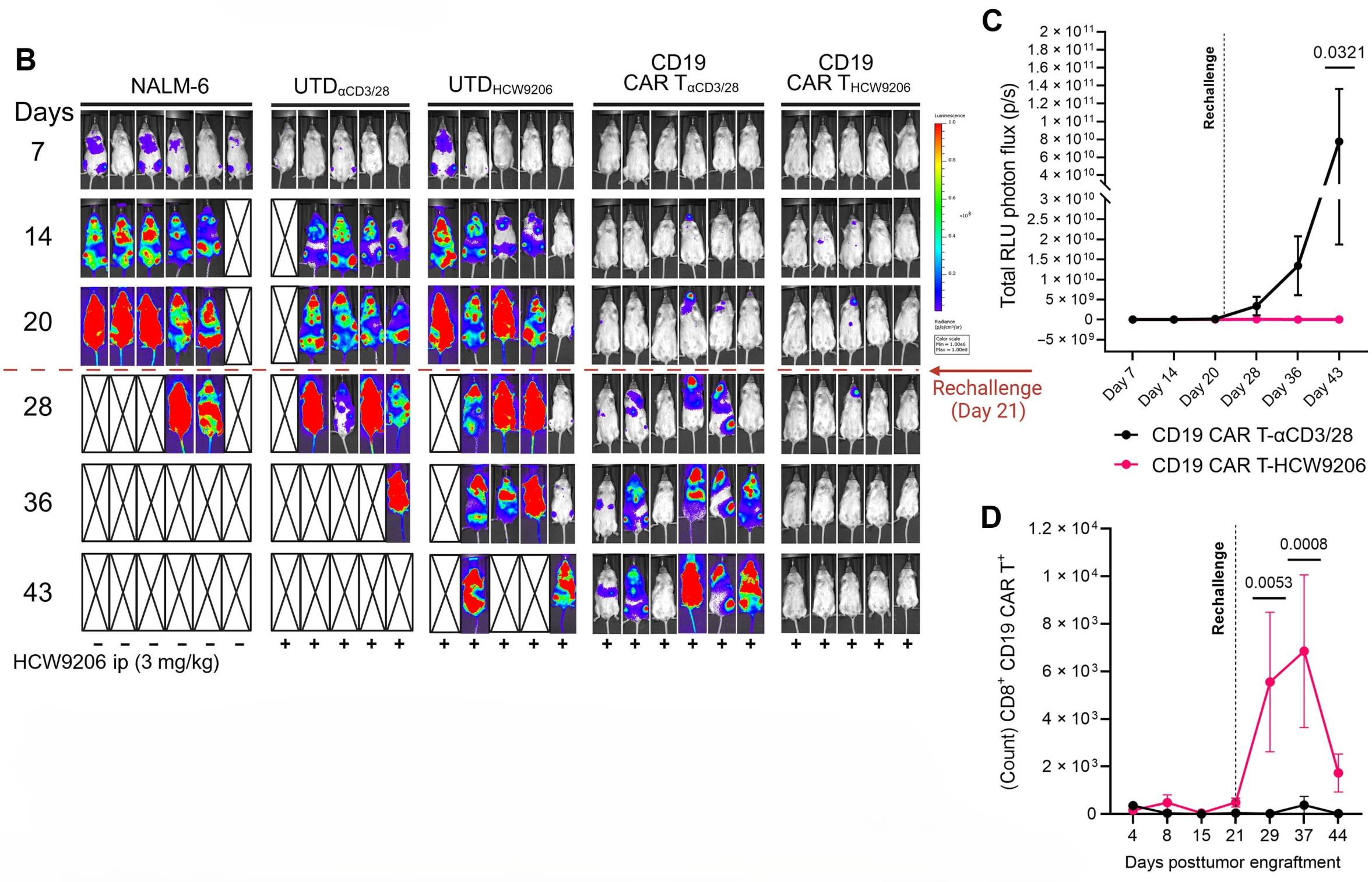
Figure 6B, C & D: In vivo tumore rechallenge model. IVIS imaging (B) and quantification (C) revealed complete suppression of NALM-6-Luc tumor expansion upon rechallenge in HCW9206-treated mice while only HCW9206-generated CAR T cells demonstrated a robust recall proliferative response in peripheral blood (D). Both the NALM-6-Luc reporter cell line and CAR T cells were generated using LentiBlast Premium. (Adapted from Cole et al., Sci Adv. 12, eaec2632 (2026).
LentiBlast Premium was used across all transduction steps in this study, from primary CD4+ and CD8+ T cells to tumor cell line engineering, demonstrating its versatility and reliability in very different cellular contexts demonstrating that it is a scalable and robust solution for lentiviral transduction T cell engineering workflow. Used simply by addition to the culture medium, without pre-coating, centrifugation or additional equipment, it delivered consistent and reproducible gene transfer across multiple donors, two distinct CAR constructs, and two manufacturing protocols.
Reference: Erin B. Cole et al. ,IL-7/IL-15/IL-21 cytokine-fusion scaffold generates highly functional CAR T cells enriched in long-lived T memory stem cells. Sci. Adv. 12, eaec2632(2026). DOI : 10.1126/sciadv.aec2632
Related Product: LentiBlast Premium
Read the article See LentiBlast Premium