BLOG > Publications & Citations > Enhancing transduction efficiency of lentiviral vectors in CAR-T cell therapy
From ~1% to ~50%: how an optimized lentiviral transduction workflow is reshaping CAR-T cell engineering
CAR-T cell therapy has transformed cancer immunotherapy, yet its clinical success depends on one essential step: the efficient delivery of CAR constructs into T cells via lentiviral vectors (LVs). While this step may appear straightforward, transduction efficiency is highly construct-specific and sensitive to a wide range of production and delivery parameters. When an anti-FITC-CAR construct yielded less than 5% transduction in Jurkat E6-1 cells using a standard protocol, Ferreira et al. set out to systematically dissect and optimize each variable of the workflow, ultimately achieving ~50% transduction efficiency, a result that underscores the transformative potential of methodical protocol refinement.
Key results
The stepwise optimization developed in this study brought transduction efficiency of the anti-FITC-CAR construct from an average of ~1% to ~50% (tdTomato⁺HA⁺ cells at 96 h post-transduction), representing a ~5–50-fold improvement depending on conditions. This was not achieved through a single intervention, but through the cumulative effect of several sequential optimizations. Among the steps tested, the addition of 1% LentiBlast Premium (total volume), without any pre-coating, centrifugation or additional equipment required, pushed the transduction from 26 to 50% in Jurkat E6-1 cells and in primary PBMCs, the optimized conditions achieved ~10% transduction at 96 h, with results maintained until day 7 post-transduction and confirmed across multiple donors.
Methodology: a sequential, modular optimization strategy
The authors applied a modular, stepwise approach, allowing the independent contribution of each variable to be assessed separately rather than modifying all parameters at once:
- LV concentration was the first and most impactful step.
- Agitation during transduction was then introduced: cells and concentrated LVs were co-incubated under agitation for 2 h at 300 rpm at 37°C. This step improved the definition of the tdTomato⁺HA⁺ double-positive population and yielded a statistically significant gain when using 150 µL of virus (1.41-fold change at 96 h, p = 0.0133), while also facilitating downstream sort-purification.
- Packaging plasmid selection was evaluated by comparing psPAX2 with R8.74 (pCMVR8.74). Although not reaching statistical significance for this construct, a consistent trend toward higher efficiency was observed with R8.74 (2.17-fold change at 96 h with 150 µL), in line with previous reports showing its superior performance in specific cell types.
- DNA input balance was then optimized by testing three envelope:packaging:transfer plasmid ratios.
- LentiBlast Premium was integrated as the final step of the workflow and added at just 1% of the total viral volume (0.5 µL for 50 µL LVs, 1.5 µL for 150 µL LVs), delivering the efficiency gains described in the next section.
LentiBlast Premium in this study
LentiBlast Premium was added at 1% of the total viral volume (0.5 µL for 50 µL LVs or 1.5 µL for 150 µL LVs) prior to the 2 h shaking incubation with cells. At this low dose, it proved effective across varying viral quantities. As shown in Figure 5, the proportion of tdTomato⁺HA⁺ cells exceeded 35% and 50% at 96 h post-transduction with 50 µL and 150 µL of anti-FITC-CAR virus respectively, compared to 17.75% and 37.77% without LentiBlast (1.36-fold change with 150 µL, p = 0.0011).
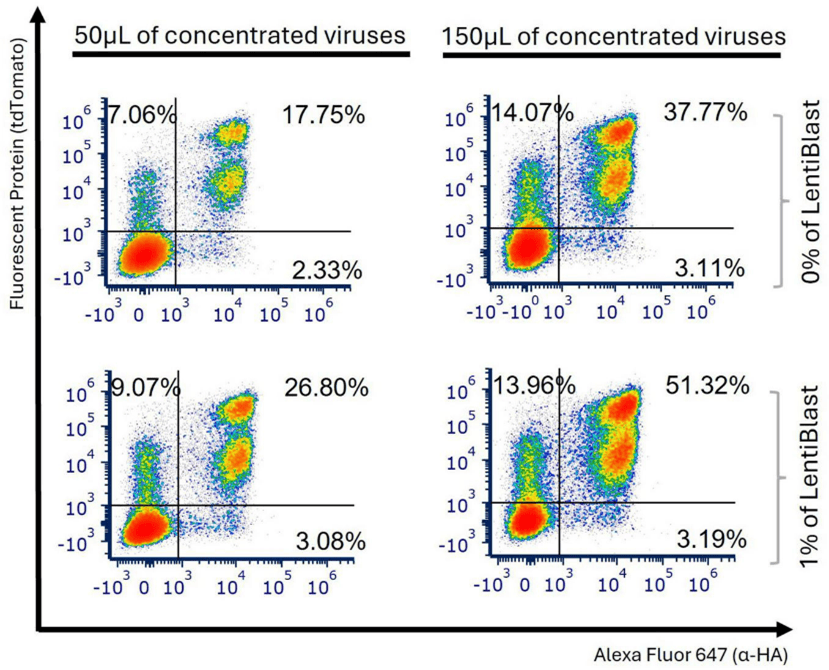
Figure 5: The efficiency of transduction was improved through the use of a transduction enhancer LentiBlast Premium. Jurkat E6-1 cells were transduced with 50 and 150 µL of anti-FITC-CAR viral particles, with shaking applied during the initial 2 h post-transduction. These viral particles were produced with R8.74 as the packaging plasmid, with a transfection ratio of 0.5:3:2. Representative plots from 3 independent experiments illustrate the levels of tdTomato and HA tag measured 96 h post-transduction, employing LentiBlast. Adapted from Ferreira et al., Front. Med. 2026, 13:1727427. CC BY 4.0.
In primary PBMCs, LentiBlast Premium significantly enhanced transduction efficiency for both the anti-FITC-CAR and WT-CAR constructs. Figure 6B illustrates the fold-change differences observed across conditions, with statistical analyses confirming the effect of LentiBlast in most conditions tested (p ≤ 0.01 and p ≤ 0.001 respectively). The authors highlight that LentiBlast Premium is a non-toxic enhancer compatible with both primary and immortalized T cells. Compared to commonly used enhancers such as Retronectin, polybrene, and dasatinib, LentiBlast Premium required no additional equipment or cell surface manipulation, offering a simpler operational workflow.
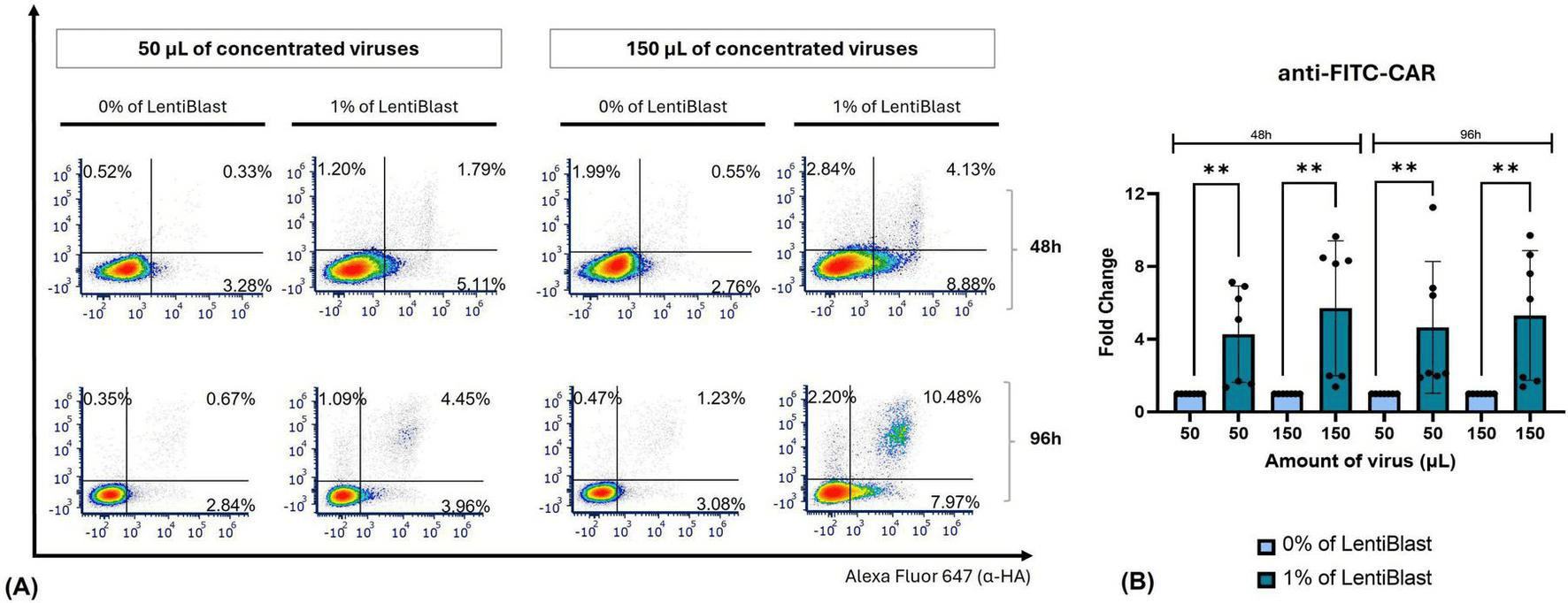
Figure 6: Fold-change differences in transduction efficiency of primary PBMCs transduced with anti-FITC-CAR concentrated viral particles, with or without LentiBlast. Data shown as mean ± SD (n = 5). ** p ≤ 0.01. Adapted from Ferreira et al., Front. Med. 2026, 13:1727427. CC BY 4.0.
The same enhancement was observed with the WT-CAR (HER2) construct in primary PBMC (figure 7) with an increase of about 76% in transduction in presence of 1% LentiBlast Premium (p≤0.001, n=5). The results illustrate that LentiBlast Premium not only allows to rescue underperforming construct but also to boot vectors that already perform well suggesting a broad-spectrum mechanism of action independent of construct-specific limitation.
Altogether the data demonstrate constant effect across two different CAR constructs, two detection markers and multiple donors, reinforcing that LentiBlast Premium is a reliable, generalized efficient transduction enhancer in T cell engineering workflows.
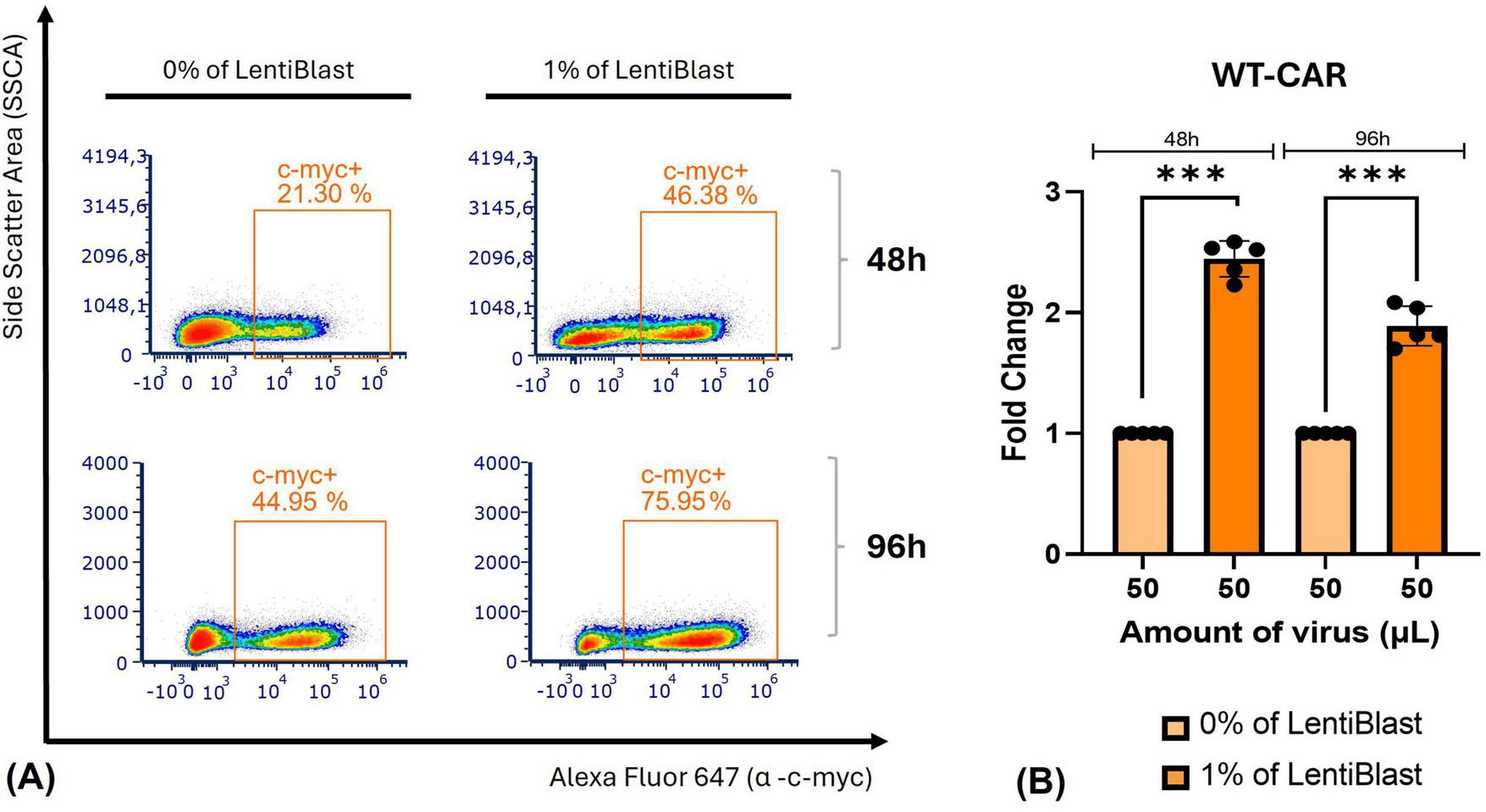
Figure 7: Transduction efficiency (A) and fold differences in transduction efficiency (B) of primary PBMC transduced with WT-CAR (HER2) non-concentrated viral particles in presence or not of 1% LentiBlast Premium. Data presented as mean ± SD (n=5). *** p≤0.001. Adapted from Ferreira et al. Front. Med. 2026, 13:1727427.
Reference: Ferreira R, Gama JFG, Godinho-Santos A and Goncalves J (2026) Enhancing the transduction efficiency of lentiviral vectors in CAR-T cell therapy through an optimization workflow. Front. Med. 13:1727427. doi: 10.3389/fmed.2026.1727427
Related Product : LentiBlast Premium
Read the article See LentiBlast Premium