No products available yet
Stay tuned! More products will be shown here as they are added.
 Custom mRNA Synthesis service
Custom mRNA Synthesis service
 Custom saRNA Synthesis service
Custom saRNA Synthesis service
 Custom LNPs service
Custom LNPs service

Latest Webinar
Want to discuss your research needs?
The RNA therapeutics have the potential to revolutionize medicinal fields as they are safe, easy to reproduce (e.g. IVT) and offer a great deal of versatility.
Naked mRNA therapeutics are generally unstable. They require high doses and show low permeability to cell membranes. Drug delivery systems are necessary to deliver mRNA for in vitro, pre-clinical and clinical applications. For this purpose, OZ Biosciences provides portfolio & custom lipid nanoparticles (LNP) delivery systems.
To date, formulation in LNP represents the most advanced non-viral delivery platform for nucleic acid therapy and promising candidates to treat manifold diseases.
LNPs are liposome-like structures, engineered for encapsulating a broad variety of nucleic acids (RNA, mRNA, siRNA, gRNA, cRNA and DNA) and APIs; LNPs consist in an aqueous core surrounded by a lipidic shell based on a combination of 4 families of chemicals, each having distinct functions (Fig.1).
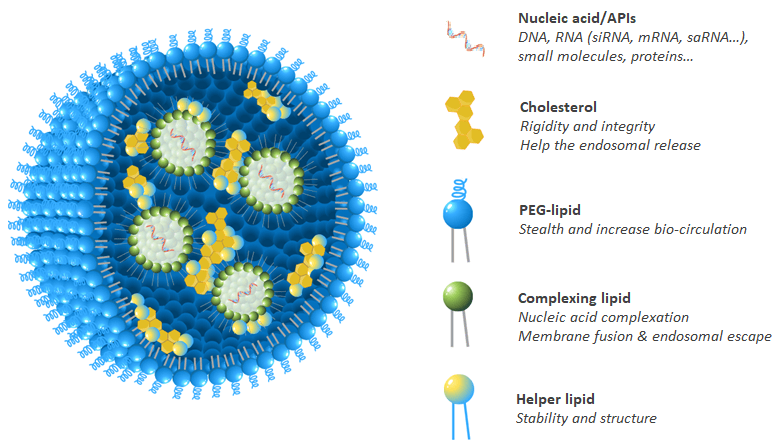
Figure 1. Schematic representation of lipid nanoparticles (LNPs) composed by a mixture of four lipidic family, usually: complexing lipid, helper phospholipid, cholesterol and stealth-lipid at defined ratio to potentiate nucleic acid activity.
In the last 20 years, OZ Biosciences has developed strong expertise in aminated lipids, which allowed the screening of several tens formulations in order to develop optimized OZ Biosciences LNPs referred as NanOZ-LNP. These proprietary lipids are protected by patents and available for licensing.
LNP/RNA systems self-assemble via electrostatic interactions between negatively charged RNA and ionizable cationic lipids. Our LNPs are produced through microfluidic technology, resulting in monodisperse nanoparticles with narrow size distribution and high encapsulation efficiency.
NanOZ-LNP have been designed as safe and advanced nanomaterials to potentiate nucleic acids/APIs activity through their effective encapsulation and delivery of payload to specific cell types and tissues.
Currently, LNPs hold great potential in diverse pharmaceutical applications including oncology, immunotherapy, regenerative medicine and chronic diseases treatment.
Formulations are available with proprietary lipids (protected by patent) or commercially available lipids (SM-102 & MC3).
Our platform can produce custom LNP formulation according to your needs and preferences.
We can support every stage of your mRNA-LNP production, from mRNA synthesis to LNP formulation development, manufacturing and fill & finish.
For any of RNA, DNA or APIs encapsulation, you can provide us with your molecule of interest and we will formulate it into LNPs.
We do know that the reproducibility of scientific experiments is very important to researchers. That is why we developed a quality control system tailored to each product, enabling us to detect defective batches immediately.
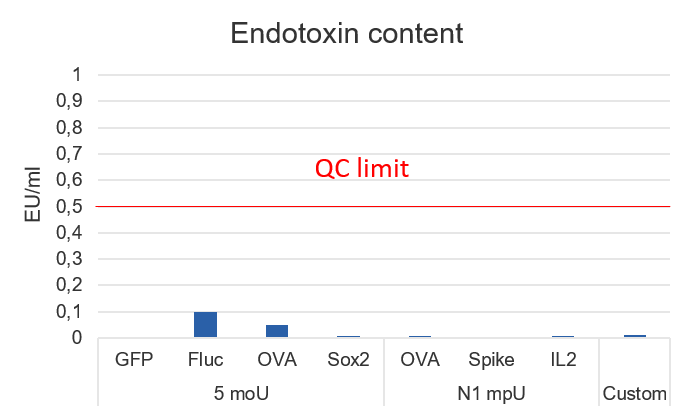
Our catalog LNPs undergo a comprehensive quality control workflow including particle size and PDI measurement by DLS, zeta potential analysis, encapsulation efficiency determination by fluorescent assay, and mRNA concentration measurement. Sterility is also assessed by incubating the LNPs in culture medium for 14 days. In addition, endotoxin detection and cholesterol content quantification are performed to further extend the analysis.
For custom LNP formulations, quality control parameters are adapted to the specific requirements of each project and formulation.
| Items | Specification | Standard QC | Superior Grade QC* |
|---|---|---|---|
| Identity | Size 50-200 nm | ✓ | ✓ |
| Charge +20mV et -20mV | ✓ | ✓ | |
| Content | Encapsulation efficiency > 80% | ✓ | ✓ |
| RNA concentration 0.1mg/mL +/-10% | ✓ | ✓ | |
| Safety | Sterility | ✓ | ✓ |
| Endotoxin <2 EU/mL | ✓ | ||
| Mycoplasma detection | ✓ | ||
| Characterization | Lipid content | ✓ |
*Our catalog LNPs undergo the standard QC. Superior Grade QC can be performed as an additional prestation.
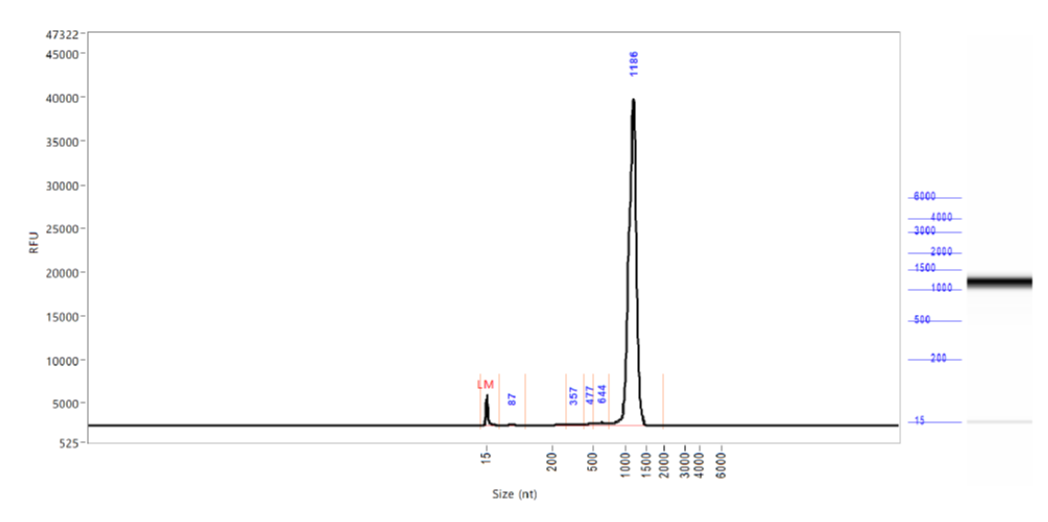
New enzyme for low dsRNA in our mRNAs!
As part of our continuous quality improvement, we have selected a new generation of RNA polymerase that reduces dsRNA during IVT. We are able to reduce the amount of dsRNA in our catalog and in our customized mRNAs by up to 88%.
Our new quality control limit is 0.05% of dsRNA.
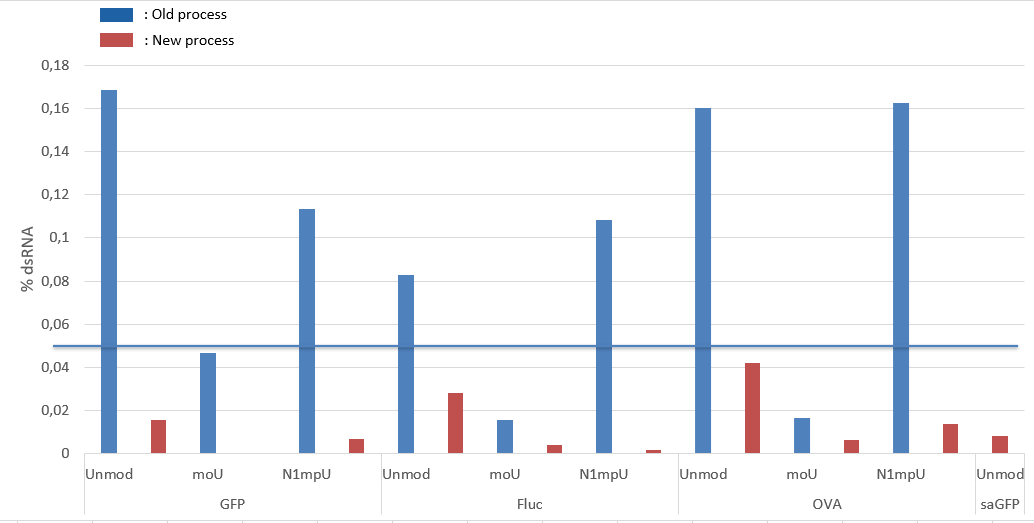
Comparison of dsRNA amount in different mRNA produced by OZ Biosciences before and after the production process improvement.
Stay tuned! More products will be shown here as they are added.